 |
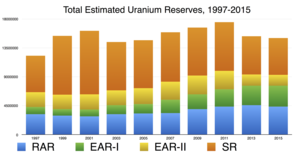
| Fig. 1: OECD world uranium reserves estimations from 1997-2015. [3] The abbreviations are defined in the text. (Source: D. Bedford) |
The term "peak uranium" describes the point at which maximum uranium production has been reached worldwide, after which peak the rate (according to the theory) will steadily decline. This concept is a highly controversial one, with much disagreement about when (if ever) this point will occur, or even whether it already has. The idea of peak uranium was proposed by M. King Hubbert in 1956, alongside similar "peak" theories concerning fossil fuels. [1] The reason for much of the controversy is that, in most cases, the fuel used for nuclear fission is uranium, and so the availability of uranium has significant implications for the entire energy industry. [2]
Since Hubbert's paper, there have been many predictions about the amount of uranium available and how long it will last. Most pessimistic predictions have been based around the fact that uranium is technically not a renewable resource; the uranium deposit on earth is finite, and it is unlikely that new uranium stores will be created in the future. [2] Estimating how much uranium actually exists on the planet is another issue in itself. Every two years, the Nuclear Energy Agency releases an updated version of the "Red Book: Uranium Resources, Production and Demand" about the state of uranium resources. [3] Its estimates are shown in Fig. 1. The acronyms are as follows:
RAR stands for Reasonably Assured Resources (RAR), which refers to "uranium that occurs in known mineral deposits of delineated size, grade and configuration such that the quantities which could be recovered within the given production cost ranges with currently proven mining and processing technology." These resources are fairly certain to exist.
EAR-I stands for Estimated Additional Resources - Category I: "uranium in addition to RAR that is inferred to occur, mostly on the basis of direct geological evidence, in extensions of well-explored deposits, or in deposits in which geological continuity has been established but where specific data, including measurements of the deposits, and knowledge of the deposits' characteristics are considered to be inadequate to classify the resource as RAR." These are thus slightly less certain.
EAR-II refers to Category II of Estimated Additional Resources, "uranium in addition to EAR-I that is expected to occur in deposits for which the evidence is mainly indirect and which are believed to exist in well-defined geological trends or areas of mineralization with known deposits."
SR stands for Speculative Resources, referring to "uranium, in addition to Estimated Additional Resources Category II, that is thought to exist, mostly on the basis of indirect evidence and geological extrapolations, in deposits discoverable with existing exploration techniques. The location of deposits envisaged in this category could generally be specified only as being somewhere within a given region or geological trend". [3]
However, these assessments may not be entirely reliable. Firstly, it must be noted that the Red Book is as much (if not more) aimed at investors as it is at scientists. Their numbers are thus based on (relatively) short-term investment, at least when compared with the timescale of peak uranium. Some variances in the graph can be explained; until 2009, all RAR estimates were of uranium that could be extracted at the price of $130/kg or less. In 2009, they added a higher price category of uranium that could be extracted at $260/kg or less. Thus, RAR estimates increase after 2009. Another interesting point is that EAR-II estimates shrink by over 50% in 2013, which is explained by the US not reporting inferred resources after 2013. The general picture in this graph is that RAR (which purports to be the most reliable estimation of resources) remains stable, even rising over the years, and while speculated amounts rise and fall, the total always adds up to about the same, even rising as well. These data can lead to some interesting conclusions; for instance, in 2009, the total of RAR and EAR-I was 5.5 million tons, and the yearly demand was 69,110 tons. This would indicate that we only have enough to last until 2089. However, the numbers for supply and demand change enough every year that such predictions are often unfounded. The amount of speculated resources has also shifted greatly over the years. [3]
There have been various proposed methods on how to avoid using up uranium resources too quickly. Fast breeder reactors showed great promise in their potential to transmute U-238 (the most plentiful isotope of uranium) into fissile Pu-239 with fast neutrons, and thus produce more fissile material than they consume. [4] They are also very appealing in their ability to extract almost all the energy from uranium fuel and to reduce both the amount and lifetime of waste produced. [5] However, these reactors have been plagued with all manner of delays, safety concerns, maintenance costs, and closures. [4,6] Although there is still promise for future designs, breeder reactor technology has not provided all that was hoped.
One alternative promising development is the extraction of uranium from seawater. Seawater contains only about 3 parts per billion uranium, but given a large enough volume, appreciable amounts can and have been extracted using polymeric absorbent fabrics. [7] When this method was in early development, there were many issues concerning its economic viability and efficiency, since costs were $138/pound of uranium, over double the spot price for uranium in U3O8 form at that time. [8] Since 2011, adsorbent uranium capacity has increased from less than 1.5 g to over 6 g of uranium per kg of adsorbent - and recently, even more strides have been made by researchers at Stanford, in the form of a new method which demonstrates a ninefold increase in uranium capacity, a fourfold faster rate of uranium accumulation, and superior reusability. [9] Given that there is an estimated 4.5 billion tons of uranium in all the world's seawater, this greatly improves the outlook for accessible uranium resources. [10]
While the estimations of the Red Book could be overoptimistic or inaccurate, at the same time, any comprehensive assessment of the uranium reserves on Earth would require inclusion of seawater in uranium, in the earth's crust, and also in any undiscovered deposits. Thus, knowing this amount for sure is nigh impossible. However, we do know that the amount of uranium (and thorium) is finite, which was the impetus for Hubbert's peak proposal and pessimistic predictions of uranium reserves. Indeed, the idea of "peak uranium" evokes images of a bleak, apocalyptic future, in which humankind has consumed all available resources and ekes out the rest of its doomed existence. Understandably, it is a controversial idea. However, regardless of the amount of uranium presently available, we must always be looking to plan ahead and secure new, renewable, and sustainable energy options. While uranium resources could potentially not be as abundant or accessible as was once thought, the view that we will simply run out of uranium and thus no longer utilize nuclear energy is pessimistic, and also does not account for the ingenuity of scientists and researchers. Discussions of this issue should not incite panic, or, on the other hand, lull people into comfort; rather, we should address the topic with honesty and the determination to make a better future.
© Dylan Bedford. The author grants permission to copy, distribute and display this work in unaltered form, with attribution to the author, for noncommercial purposes only. All other rights, including commercial rights, are reserved to the author.
[1] M. K. Hubbert, "Nuclear Energy and the Fossil Fuels," Shell Development Company, Publication No. 95, June 1956.
[2] N. Chowdhury, "Is Nuclear Energy Renewable Energy?" Physics 241, Stanford University, Winter 2012.
[3] "Uranium 2014: Resources, Production and Demand," Nuclear Energy Agency, NEA No. 7209, 2014. [Also versions of this publication for the years 1999, 2001, 2003, 2005, 2007, 2009, and 2011.]
[4] I. Waisberg, "The Integral Fast Reactor (IFR) - An Unfulfilled Promise," Physics 241, Stanford University, Winter 2013.
[5] C. Jones, "The Basics of Integral Fast Reactors," Physics 241, Stanford University, Winter 2015.
[6] S. Abdul-Kafi, "The Superphenix Fast-Breeder Reactor," Physics 241, Stanford University, Winter 2011.
[7] N. Seko et al. "Aquaculture of Uranium in Seawater by a Fabric-Adsorbent Submerged System," Nucl. Technol. 144, 274 (2003).
[8] B. Chan, "Amidoxime Uranium Extraction From Seawater," Physics 241, Stanford University, Winter 2011.
[9] C. Tsouris. "Uranium Extraction: Fuel From Seawater," Nat. Energy 2, 1 (2017).
[10] N. F. Lane et al., "Powerful Partnerships: The Federal Role in International Cooperation on Energy Innovation," Office of the President of the United States, June 1999.