 |
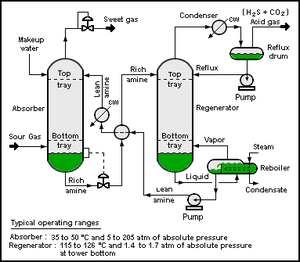
| Fig. 1: A rough outline of the Amine Treating process. "Sour" gas contains the substances to be filtered. "Sweet" gas has been filtered. "Rich" amine has the contaminants dissolved within, while "lean" amine does not. (Source: Wikimedia Commons) |
As climate models suggest that the continuing bulk emission of carbon dioxide (via the burning of fossil fuels) will continue to exacerbate climate issues, alternative sources of energy have become increasingly sought-after. However, fossil fuels remain the most economical energy source, both because the technology to create energy from them is older and more developed, and because of their relatively high energy content. As such, technologies to reduce atmospheric carbon dioxide emissions by capturing and storing carbon dioxide from fossil fuel combustion have been an increasingly popular point of discussion.
The current dominant method of carbon capture is via amines, which have been used to remove carbon dioxide in industrial applications for decades. [1] The technique involves aqueous amine solutions which absorb acidic gasses, including carbon dioxide (which forms carbonic acid in water) (Fig. 1). The amine-rich solution is poured downward onto a rising gas flow, allowing the desired gasses to react with the amines and be absorbed into the solution; the solution is then reheated in a separate chamber, releasing the captured gasses into containment.
The captured gas then must be stored. Currently, it is usually transported via pipeline for various industrial purposes, and transport for sequestration via pipeline would thus be a simple adjustment. [2] However, storage is a somewhat more difficult problem. Ocean storage was suggested in the past, but poses obvious problems due to the aforementioned acidic tendencies of carbon dioxide. [2] The most prominent proposed storage space is in geological formations, with the gas often being pumped into emptied oil or gas pockets under high pressures (150 bar). [2,3]
However, energy requirements of this process are non-negligible. For coal, for instance, the theoretical minimum energy to extract carbon dioxide from the flue gas and pressurize it for efficient storage (150 bar) is 396k J/kg of carbon dioxide, though the lowest energy claimed to be achievable is closer to 720 kJ/kg. [1] Given typical coal consumption yields are 3.65 MJ/kg carbon dioxide, this means carbon sequestration for coal would require at least 10% of the produced energy to be diverted to the sequestration process, and in reality would divert 20% or more, making carbon sequestration a fairly expensive proposition. [4] This calculation totally neglects the energy costs of pumping the carbon dioxide to the storage point, which could raise costs substantially for sufficiently remote storage locations.
Furthermore, as carbon dioxide will never be fully absorbed into the amine solution, the process can, at most, absorb 85-90% of the emitted carbon dioxide; assuming extra coal is burned to compensate for the energy cost of sequestration, this would mean that carbon dioxide emissions from these power plants would remain at at least 12.5-18.75% of original levels should all coal plants be fitted with the these technologies, and likely higher assuming not all plants were able to achieve the lowest-level 20% energy consumption. Conversion to natural gas, with a yield of 6.50 MJ/kg carbon dioxide, would reduce carbon dioxide emissions of the affected power production by 44% compared to conventional coal. [4] While this is roughly half of the potential largest emissions reduction for equipping all coal plants, it comes without any required technology development and in a form that is already economically viable, as is demonstrated by the large number of natural gas power plants currently in operation. [4]
While carbon capture technology could potentially reduce carbon emissions from coal consumption significantly, it would also likely render coal-based energy significantly more expensive; this, in turn, would likely render coal fairly uncompetitive economically. Switching even to natural gas would substantially reduce carbon emissions without increasing the cost per unit energy anywhere near as dramatically, as natural gas plants are currently economically feasible. Carbon capture is thus technically achievable, but of limited economic feasability.
© Matthew Sorensen. The author grants permission to copy, distribute and display this work in unaltered form, with attribution to the author, for noncommercial purposes only. All other rights, including commercial rights, are reserved to the author.
[1] G. T. Rochelle, "Amine Scrubbing for CO2 Capture," Science 325, 1652 (2009).
[2] W. Chen, "Carbon Capture and Sequestration," Physicsc 240, Stanford University, Fall 2010.
[3] K. S. Lackner, "A Guide to CO2 Sequestration," Science 300, 1677 (2009).
[4] V. Krey et al., "Annex II: Metrics and Methodology," in Climate Change 2014: Mitigation of Climate Change. Contribution of Working Group III to the Fifth Assessment Report of the Intergovernmental Panel on Climate Change, ed. by O. Edenhofer et al. (Cambridge University Press, 2014), p. 1307.