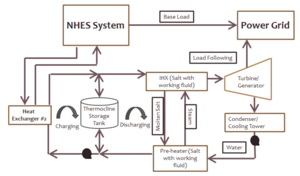
 |
| Fig. 1: Thermocline molten salt storage for Nuclear Hybrid Energy System (NHES). [2] (Courtesy of the U.S. Department of Energy) |
The realities of renewable energy sources are well known: the sun doesn't shine at night and the wind doesn't always blow on cue. Natural fluctuations in renewable power sources do not generally coincide with electricity demand resulting in a mismatch of input and output. Unfavorable weather only exacerbates the problem since turbines won't operate continuously at maximum efficiency. Nuclear power suffers from a similar problem since the reactors cannot accommodate steep increases in demand, or "ramping". Even without fluctuations, regulation ramping requirements, such as the recommendation that California ISO systems are able to provide ramps between 40 - 60 MW per minute for up to 6 minutes, compound the difficulties faced by non-carbon power sources. [1] Cost effective methods for large-scale energy storage would therefore result in a more flexible power grid by absorbing surplus energy during off-peak hours, mitigating the ramping problem, and smoothing out the capricious renewables.
There are a number of different means to store energy including mechanical, electrical, and chemical as shown in Table 1. These storage methods are high quality since it is possible to directly convert energy from one form to the other. To minimizing cost, energy storage systems should maximize energy density and charging rates while minimizing losses and leakage. [2] For large-scale storage, chemical and electrical methods suffer from energy losses. Mechanical storage, such as hydropumping and compressed air, has a low energy density and depends on geographic location. The last type of energy storage is thermal, which is low quality in the sense that there are fundamental thermodynamic costs in converting thermal energy to electrical energy. While perfect energy efficiency cannot be achieved, in real-world applications we are only concerned with economic efficiency such that the cost calculus can make thermal storage a viable option.
|
||||||||||||||||
| Table 1: Different types of energy storage. [2] |
Thermal storage stores energy in the form of heat that is either "sensible" or "latent". Sensible heat corresponds to thermal storage in a single phase where the temperature of the material varies with the amount of stored energy. [2-4] The equation for heat flow from hot to cold is: Q = m C ΔT where Q is the (sensible) heat, m is the mass, C is the specific heat, and ΔT is the temperature difference. Both liquid (water, oil, molten salt) and solid (rock, metal) media can be used. [4] This requires insulation to maintain a temperature gradient to store the heat. [3] For low temperature (25 - 90°C), water is ideal; it has a relatively high specific heat (4.2 kJ/kg°C), is cheap, and over a 60°C temperature difference can store 250 kJ/kg. [4] Operating at low temperatures avoids complications associated with the high vapor pressure and containment. Latent heat is stored using phase-change materials that take advantage of the (specific) latent heat L associated with a phase transition: Q = m C ΔT + m L though this is not as efficient at high temperatures. [3] Phase-change materials are appealing because of their high energy storage density; water, for example, has a latent heat of fusion 335 kJ/kg which is approximately 80 times the amount of heat that is required to raise the temperature of 1 kg of water by 1°C. [4] While latent heat may be suitable for a house with solar panels, additional complications associated with cost and repeated use currently make sensible heat a better option for large-scale storage. [5]
 |
| Fig. 2: Solar Two thermal power plant. (Source: Wikimedia Commons) |
Molten salts have high boiling points, low viscosity, low vapor pressure, and high volumetric heat capacities. [2] A higher heat capacity corresponds to a smaller storage tank volume. In choosing the chemical mixture, it is advantageous to have the lowest possible melting point (and highest boiling point) to maximize the available temperature range for the molten salt. If the melting point is too high, additional heating may be required to prevent freezing. [2] Salts used for storage (such as sodium nitrate NaNO3 and potassium nitrate KNO3) have melting points between 300-500°C and volumetric heat capacities between 1670 - 3770 kJ/m3°C. [2] Sodium hydroxide NaOH has a melting point of 320°C and can be used up to 800°C, but is highly corrosive. [4] Commercially available "HITEC" salt used in solar plants consists of potassium nitrate (53% by weight), sodium nitrite NaNO2 (40% by weight), and sodium nitrate (7% by weight) with a liquid temperature range of 149 - 538°C. [6]
The salts are heated and stored in an insulating container during off-peak hours. When energy is needed, the salt is pumped into a steam generator that boils water, spins a turbine, and generates electricity. The conversion of thermal energy to electricity can proceed by different cycles such as the Rankine, Brayton, and Air-Brayton cycles. [2] The Brayton gas cycle, for example, involves (1) adiabatic expansion of the high-pressure and high-temperature gas across a turbine to do work, (2) isobaric cooling, (3) adiabatic compression to high pressure, and (4) isobaric heating whereupon the cycle is repeated. Other applications include using the stored heat directly for high temperature processes (such H2 production and coal-to-liquid conversion), which avoids the thermodynamic cost incurred from converting to electricity. [2] The cooled salt is pumped back into the storage tank to be heated and reused.
There are two different configurations for the molten salt energy storage system: two-tank direct and thermocline. The two-tank direct system, using molten salt as both the heat transfer fluid (absorbing heat from the reactor or heat exchanger) and the heat storage fluid, consists of a hot and cold storage tank. [2] The thermocline system (see Fig. 1) uses a single tank such that hot and cold salt are separated by a vertical temperature gradient (due to buoyancy force) to prevent mixing. [6] There are two cycles in the thermocline system: charging and discharging. To charge, salt flows out of the cold side, is heated by the heat exchanger (reactor), and flows into the tank's hot side. To discharge, salt flow out of the hot side, transfers heat to generate power (turbine), and flows into the tank's cold side. [2,6,7] The thermocline system reduces costs through a single tank and cheap filler material in the tank to act as thermal storage; the estimated cost relative to the two-tank direct system is about 35%. [6,7]
Thermal energy storage is currently being used in concentrated solar plants consisting of parabolic mirrors (troughs) or sun-tracking mirrors (heliostats) that direct sunlight at a focal point receiver tube in the trough or a single "power tower" (shown in Fig. 2) where a heat transfer fluid (typically petroleum based oils) absorbs the energy. [8] Oils have a lower vapor pressure than water and low melting points; they can operate at temperatures above 300°C, but are impractical for heat storage above 400°C due to high vapor pressure. [6] The heated oil can be directed to a steam generator for electricity, or a heat exchange can transfer the energy to molten salts to be stored (with round-trip efficiencies of 95 - 99%). [5,9,10] To eliminate this intermediate exchange in indirect plants, direct plants use molten salt, which operates at low vapor pressure at 500°C, as the heat transfer fluid; this has the added benefit that molten salts are cheaper and more environmentally friendly than synthetic oil, but have higher melting points and therefore may require freeze protection. [6] The 10 MW retrofitted "Solar One" tower plant, "Solar Two" (decommissioned in 1999), in the Mojave Desert was the first to demonstrated the direct plant feasibility using molten salt as a heat transfer fluid. [5] The mixture, consisting of 60% sodium nitrate and 40% potassium nitrate, melts at 220°C. "Cold" molten salt at 260°C is then heated to about 550°C and stored. [5,11]
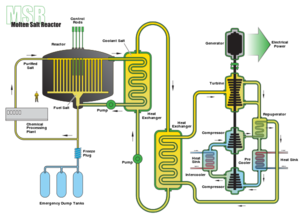 |
| Fig. 3: Solid fuel molten salt nuclear reactor schematic. (Source: Wikimedia Commons) |
In 2008, the "AndaSol-1" solar thermal plant in Spain became the first commercial parabolic trough plant in Europe. AndaSol-2 and AndaSol-3 followed soon after in 2009 and 2011 using two-tank indirect storage systems that hold 28,500 tons of molten salt. [2] The organic heat transfer fluid is a mixture of diphenyl ether C12H10O and biphenyl C12H10, which melts at 12°C and operates up to 390°C. The three 50 MW plants can store up to 1010 MWh of energy in molten salt via a heat exchanger with a storage capacity of 7.5 hours. [2,5] There are currently four solar thermal plants with outputs of 250 - 392 MW operating in U.S. where solar provides only a few percent of renewable energy consumption and about 0.2% of the total energy consumption. [11,12]
While these plants serve as proof of concept for large-scale molten salt technology, the rarity of commercial solar thermal storage comes down to cost. The estimated cost for a hypothetical 200 MW molten salt power tower is $30/kWh and $200/kWh for a synthetic oil parabolic trough plant. [10] The capital cost of a 688 MWh two-tank molten salt storage system was similarly estimated to be $31/kWh. Improvements using a thermocline system with quartzite rock filler were estimated to bring the cost down to $20/kWh. [7] We can get a very rough estimate on the price per kWh of the AndaSol plant by considering the capacity factor: the average power produced over a 24 hour cycle divided by the full capacity output. The capacity factor improvement with storage can provide an indirect measure of the fractional cost of the storage system. For solar, the capacity factor is limited to 20 - 25% without storage and up to 50% (troughs) and 77% (power tower) with storage. [9] Assuming a factor of three improvement in the capacity factor and a cost of $345 million (315 million Euros) for AndaSol, our crude estimate is:
| Storage Cost | = | $345,000,000 3 x 1,010 MWh |
1 MWh 1,000 kWh |
= | $114 / kWh |
This indirect determination overestimates the cost since it doesn't take into account the ability of thermal storage to sell electricity during peak demand hours.
With the inevitable end of the carbon based fuels, either by law or by burning to completion, it is important to consider other fuels with significant lifetimes. While nuclear fission is not a "renewable" fuel source, estimates from the current burn rate suggest it will last for a few hundred years and, with advanced breeding techniques, can sustain the world's energy budget for over a thousand years. [13] It therefore makes sense to study the application of molten salt to nuclear power and the cost calculus to serve as a possible base load source using thermal storage when carbon fuel is depleted. The technology for using molten salt in nuclear reactors is not new. In 1954, the U.S. aircraft reactor experiment operated a molten salt reactor (MSR) for 100 hours at temperatures up to 860°C and powers up to 2.5 MW. [14] Oak Ridge National Laboratory operated a 7.4 MW test reactor from 1965 - 1969 and demonstrated the viability of different liquid fuel systems. [15]
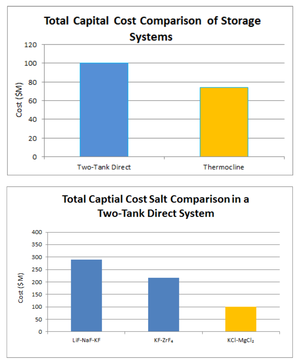 |
| Fig. 4: Capital cost estimate (million dollars) for 300 MWh molten salt storage system. [2] (Courtesy of the U.S. Department of Energy) |
These nuclear fission reactors can use a molten salt mixture as either the low-pressure coolant for a solid fuel reactor (see Fig. 3) or as liquid fuel by dissolving fissile material in the salt. [14,15] The advantage is a higher operating temperature (700-750°C) and lower vapor pressure than light water reactors. Moreover, fuel rods don't need to be created for liquid fuel reactors. The fissile fuel, dissolved in the molten fluoride coolant as uranium tetrafluoride UF4, undergoes fission in the salt and flows to a heat exchanger to transfer the heat to a secondary molten salt coolant. [14,15] The choice of salt depends on its tendency to capture neutrons and ability to slow down neutrons (moderate); LiF and BeF2 mixtures are common since the salt has both low neutron capture and is an effective moderator that increases fission cross section. [15] One safety feature of the liquid fuel MSR is the thermal expansion that slows down the rate of reactions to create a negative feedback loop. [14] Additionally, the salt can be continuously processed to remove fission products. For breeding in a "two-fluid system", a thorium salt blanket around the fissile uranium salt fuel captures neutrons. The generated U-233 is removed by fluorination whereby fluorine gas is bubbled through the blanket turning UF4 to volatile UF6. [14] The UF6 is collected and turned back to UF4 to be added to the liquid fuel salt. The two-fluid system has the advantage of separating the removal of rare earth fission products (that absorb neutrons and inhibit breeding) from collection of thorium, which has similar chemical properties. [14] The drawback is a more complicated vessel to separate the fluids. The "single-fluid system" used in the 7.4 MW Oak Ridge 1965 MSR Experiment avoided this complication and only used fissile fuels U-233, U-235, and Pu-239. [14]
The heat exchange with the secondary molten salt for storage may offer a cost advantage for MSR and large-scale thermal storage to adjust to electricity demand fluctuations. An recent analysis of the nuclear/molten salt storage combination found a thermocline system (with Air-Brayton combined power cycle) was the most economically efficient and the two-tank system was the most thermally efficient design. [2] Thermal efficiency depended on the power cycle more than salt choice whereas the opposite was true for economic efficiency. While LiF-NaF-KF salt yielded the highest thermal efficiency due to its high volumetric heat capacity (and therefore low rate of heat loss in the storage container), KCl-MgCl2 salt resulted in a significantly lower capital cost (see Fig. 4). For a 300 MWh thermocline system with KCl-MgCl2 salt, the capital cost is estimated to be about $75 million corresponding to a cost of $250/kWh.
Lastly we would like to compare the cost of molten salt with another large-scale storage method: hydropumping. The principle of operation behind hydropumping is as simple as it sounds: pump water uphill to store gravitational potential energy for use later by driving a turbine on its way back down. While there exists weekly and seasonal variations in demand (highest being winter and summer), typical storage capacities are around 10 hours to level loads; this follows because there is no compensation for storing energy, and the most revenue is therefore generated by charging (when the price of electricity is low) and discharging (when the price is high) with maximum frequency. [2] Losses from evaporation and leaking still produce a round-trip energy efficiency of about 80%. [16] In terms of economies of scale, hydropumping can store over 129 GW worldwide. [16]
|
|||||||||||||||||||||||||||||||||||
| Table 2: U.S. hydropump facilities. [17] |
In Table 2 we have listed U.S. hydropumping facilites with their power, capacity, capital cost in inflation adjusted dollars (to the year 2000), and the cost per kWh. [17] The storage costs are all less than $100/kWh with the lowest at $14/kWh; this is clearly less than molten salt coupled to either nuclear power or solar power. The main drawback of hydropumping is the dependence on geographic location since the reservoirs are typically built into the topography. Not only does hydropumping take up a lot of space, but there will also be more evaporation (and much less water) in regions ideal for solar plants, i.e. the desert. Molten salt is therefore an option when geography prevents hydropumping and requires higher energy density storage.
Molten salt can function as a large-scale thermal storage method that would allow other energy sources, such as nuclear and solar, to become more feasible by smoothing out the fluctuations in demand and weather. The lack of widespread use comes down to costs; hydropumping is less expensive where applicable, and renewable sources don't account for enough of the energy consumption pie to implement thermal storage. Molten salt nuclear reactors may continue to grow, but more developments are needed before large-scale thermal storage for nuclear power becomes competitive. Looking to the future after depleting carbon-based fuel, the successful demonstration of molten salt storage for solar power will provide a price floor for tapping into the desert to meet our power demands.
© John Dodaro. The author grants permission to copy, distribute and display this work in unaltered form, with attribution to the author, for noncommercial purposes only. All other rights, including commercial rights, are reserved to the author.
[1] Y. V. Makarov, et al. "Assessing the Value of Regulation Resources Based on Their Time Response Characteristics," PNNL-17632, June 2008.
[2] M. Green, et al. "Nuclear Hybrid Energy Systems: Molten Salt Energy Storage," INL/EXT-13-31768, November 2013.
[3] B. Reinhardt, " Thermal Energy Storage," Physics 240, Stanford University, Fall 2010.
[4] S. M. Hasnain, "Review on Sustainable Thermal Energy Storage Technologies, Part I: Heat Storage Materials and Techniques," Energy Convers. Manage. 39, 1127 (1998).
[5] C. Barile, "Solar Thermal Energy Storage Systems," Physics 240, Stanford University, Fall 2010.
[6] Z. Yang, and S. V. Garimella, "Cyclic Operation of Molten-Salt Thermal Energy storage in Thermoclines for Solar Power Plants," Appl. Energy 103, 256 (2013).
[7] J.E. Pacheco, et al. "Development of a Molten-Salt Thermocline Thermal Storage System for Parabolic Trough Plants," J. Solar Energy Eng. 124, 153 (2002).
[8] Y. Rajavi, "Concentrating Solar Power," Physics 240, Stanford University, Fall 2013.
[9] S. Herron, "Solar Irradiation and Energy from Deserts," Physics 240, Stanford University, Fall 2010.
[10] E. A. DeMeo and J. F. Galdo, "Renewable Energy Technology Characterizations," Topical Report TR-109496, US Department of Energy, December 1997.
[11] A. Blandino, "Solar Thermal Energy," Physics 240, Stanford University, Fall 2014.
[12] "Annual Energy Review 2011," DOE/EIA-0384(2011), September 2012.
[13] R. Wilson, "The Changing Need for a Breeder Reactor," Nucl. Energy 39, 99 (2000).
[14] J. Serp, et al. "The Molten Salt Reactor (MSR) in Generation IV: Overview and Perspectives," Prog. Nucl. Energy 77 308 (2014).
[15] Y. Kelaita, "Molten Salt Reactors," Physics 241, Stanford University, Winter 2015.
[16] M. Jiang, "Large Scale Energy Storage," Physics 240, Stanford University, Fall 2011.
[17] O. Galvan-Lopez, "The Cost of Pumped Hydroelectric Storage," Physics 240, Stanford University, Fall 2014.