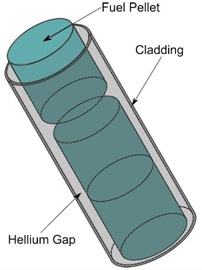
 |
| Fig. 1: Simplified schematic of a fuel rod, comprised of three components: fuel pellets, enclosed in a protective cladding, separated by a small gap that is filled with helium gas. |
With the world trying to move away from its traditional fossil fuel resources, the nuclear power industry is posed to grow if the economics of implementation and the reliability of the technology exist. Increases in efficiency of nuclear fuel usage help promote nuclear power adoption, and there exists a large body of research around the subject. Recently, International Beryllium Corporation (IBC) Advanced Alloys have undertaken collaborative research with Purdue University and Texas A&M University to develop a new type of longer lasting, more efficient and safer beryllium oxide nuclear fuel.
A simplified figure of a nuclear fuel rod is shown below (Fig. 1). Fuel pellets are typically formed by pressing and sintering uranium dioxide (UO2) into fuel pellets. UO2 has poor thermal conductivity, resulting in a wide temperature gradient through the pellets, with the highest temperatures being reached in the center of the pellet. UO2 expands in response to these high temperatures, thus closing the helium gaps that are illustrated in Fig. 1. Additionally, the rate at which reaction gases build up are increased, consequently increasing the pressure and causing these fuel rods to fail.
Introducing a new material is done with the intention of increasing the poor thermal conductivity. Beryllium oxide (BeO) is selected as the primary candidate because BeO has best thermal conductivity of any oxide along with its non-reactiveness with UO2 at temperature levels lower than 2100°C. [1] The first phase of research involved nuclear engineering simulations and thermal modeling. The results showed the same temperatures at the surface of the pellets but decreased temperatures at the centerline - overall, reducing the average temperature of the pellets. Additional effects include the fuel pellet's increased reactivity, burn-up, and lifespan. These kinds of effects could be enormously beneficial in longer lasting nuclear fuel (increase in fuel efficiency) and less frequent refueling (lesser maintenance and refueling costs).
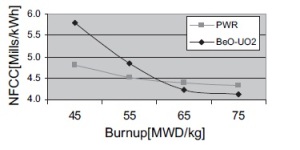 |
| Fig. 2: Break-even point between BeO-UO2 fuel and UO2 fuel, as a function of nuclear fuel cycle cost (NFCC) and burnup (MWD/kg). [2] |
However, no newly implemented technology comes without its costs, better known as the no-free-lunch principle in economics. An extensive cost-benefit analysis of using BeO-UO2 indicates the dominant cost-benefit drivers of BeO-UO2 fuel: [2]
Decrease of nuclear fuel cycle cost/increased lifespan of the fuel, due to the higher thermal conductivity and its resulting higher burn-up.
Decrease of disposal costs due to lesser radioactive waste generation and longer lifetime of nuclear fuel.
The cost reduction of direct materials by reusing Be; the so-called "Be Credit".
Higher prices of beryllium, as this material is more expensive than uranium.
Higher cost of manufacturing, as the process to mix Be-O with UO2 involves a relative high degree of complexity.
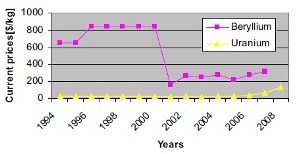 |
| Fig. 3: Cost of beryllium and uranium in the past 16 years. [2] |
Using beryllium oxide in the nuclear fuel presents a tradeoff between increased performance for the reasons stated above and the costs associated with the technology. In addition, these parameters are subject to the constraint that the content of BeO must satisfy safety requirements, as BeO content can affect the corrosion of the fuel tube and the pressure between the pellet and the tube of the nuclear fuel. In conducting the cost-benefit analysis, the optimal BeO content was determined to be 4.8% by weight (operating under the assumption that BeO and UO2 were priced at $317/kg and $64/kg, respectively). [2] Additionally, when considering the nuclear fuel cycle cost (NFCC) as a function of the burn-up, the BeO-UO2 fuel is only economically efficient when the burn-up is more than 60 MWD/kg, shown in Fig. 2. [2]
Even though an industrial scale solution to create this mixture in large quantities has yet to be developed, the cost driver that plays the largest role is the disparity between the price of beryllium and uranium (Fig. 3). The price gap between the two materials narrowed significantly since the turn of the century, but as demand for beryllium increases, the gap can once again grow large. If this price gap increases, the economic benefit of is largely diminished.
© Mehran Nazir. The author grants permission to copy, distribute and display this work in unaltered form, with attribution to the author, for noncommercial purposes only. All other rights, including commercial rights, are reserved to the author.
[1] K. H. Sarma et al., "New Processing Methods to Produce Silicon Carbide and Beryllium Oxide Inert Matrix and Enhanced Thermal Conductivity Oxide Fuels," J. Nucl. Mat. 328, 6 (2006).
[2] S. K. Kim