 |
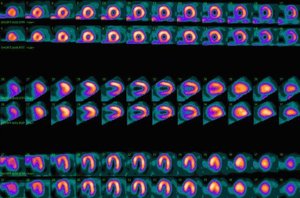
| Fig. 1: Myocardial perfusion scan. (Source: Wikimedia Commons) |
Myocardial perfusion scans are non-invasive imaging tests that allow physicians to see how well blood flows through heart muscle, also known as the heart's perfusion (see Fig. 1). [1] In doing so, it can show how well the heart muscle is functioning to pump blood and it can show which areas of the heart arent receiving proper blood flow both during rest and during physical stress such as exercise. [1] In order for the scan to work, subjects are administered tracers, which are small amounts of radioactive material that mix with blood, allowing them to be taken up by the heart muscle as the blood flows through arteries. [1] A special gamma camera then takes images of the heart, allowing physicians to determine the perfusion within specific areas of heart muscle. [1] Myocardial perfusion scans can be obtained using either single photon emission computed tomography (SPECT) or positron emission tomography (PET). [2]
The radioactive tracers used from perfusion imaging with SPECT are Tl-201 and two Tc-99m. [2] Tl-201 decays to Hg-201 by electron capture, emitting both X-rays and gamma photons. [2] Thallium is administered intravenously as thallous chloride, with 88% of it being cleared from the blood after one circulation, and 4% of the tracer localizing in the myocardium. [2] The thallium then enters the cardiac myocytes through either the sodium-potassium pump or down the cells electrochemical gradient. [2] The tracer is distributed in the myocardium based on the myocardial perfusion and viability. [2] Because Tl-201 redistributes from its initial distribution over several hours, images to reflect baseline perfusion and viability can be acquired three to four hours after intravenous administration. [2] Tl-201 is a good clinical tracer, but because it has a relatively long physical half-life, it has a high radiation burden for the patient. [2] It also has a relatively low injected activity and low energy emission, causing significant attenuation and creating a low signal-to-noise ratio, both of which make it more difficult to produce high quality images. [2]
Because of these limitations, Tc-99m is also used as a tracer in SPECT despite its lower fractional myocardial tracer uptake. The two technetium-99m tracers currently available are Tc-99m-sestamibi and Tc-99m-tetrofosmin. [2] Tc-99m-sestamibi is retained based on intact mitochondria, and can diffuse through the capillary and cell membrane, but not as easily as Tl-201, decreasing the initial extraction. [2] The kidneys and hepatobiliary system are responsible for eliminating most of the Tc-99m-sestamibi. Tc-99m-tetrofosmin is also cleared rapidly from the blood, resulting in a myocardial uptake similar to that of Tc-99m-sestamibi. [2] The kidneys and hepatobiliary system are responsible for eliminating most of the Tc-99m-sestamibi, but Tc-99m-tetrofosmin hepatic clearance is slightly more rapid than in the case of Tc-99m-sestamibi. [2] Both technetium-99m tracers are taken up by cardiac myocytes and are distributed within the myocardium based on myocardial perfusion, but they have little to now redistribution so two separate administrations of Tc-99m are necessary in order to get both stress and resting images. [2] Additionally, myocardial perfusion rate is more closely related to the uptake of Tl-201 compared to the uptake of Tc-99m tracers, so defects in myocardial perfusion may not be as easily distinguishable. [2] However, Tc-99m tracers have higher energy and less attenuation, which allows for higher image quality. [2] They also have shorter half-lives, allowing for larger administrations with higher activity levels. [2]
There are several tracers available for clinical cardiac PET imaging, such as N-13-ammonia, O-15-water, and Rb-82, but they are often used in combination with F-18-FDG. [2] F-18-FDG is a metabolic tracer and is the most common tracer used for clinical cardiac PET scans. [2] It is a metabolic tracer because glucose uptake results in FDG clearance in the blood, so high blood plasma concentrations degrade the quality of the images. [2] FDG uptake in the myocardium increases with regional glucose usage such as myocardial work. [2] This method is used for patients with dysfunctional myocardial regions due to hypoperfusion because it can determine the likelihood or benefit of revascularizationif the PET scan with FDG shows residual metabolic activity, then that region of the dysfunctional myocardium is viable with the potential of functioning again. [2] However, if the image indicates decreased regional FDG uptake relative to myocardial perfusion, then the myocardium is likely permanently damaged. [2]
After administration of the tracers, gamma cameras which are external detectors of radiation are able to create images from the radiation emitted by the tracers. [2] Through these images, clinicians are able to determine whether the heart muscle is receiving enough blood, or if blood flow to certain parts of the heart are reduced, which usually indicates blocked or narrowed arteries. [3] Myocardial perfusion scans can also indicate scarring of the heart muscle due to previous cardiac events such as heart attacks since scars are dead tissue, or they can determine whether the regions of heart muscle that are damaged due to heart attacks are permanently damaged, or if the heart muscle will still be viable after bypass surgery or after placement of a coronary stent. [3]
© Catherine Raquel. The author warrants that the work is the author's own and that Stanford University provided no input other than typesetting and referencing guidelines. The author grants permission to copy, distribute and display this work in unaltered form, with attribution to the author, for noncommercial purposes only. All other rights, including commercial rights, are reserved to the author.
[1] J. C. Lee, M. J. West, and F. A. Khafagi, "Myocardial Perfusion Scans," Aust. Fam. Physician. 42, 564 (2013).
[2] B. Hesse et al., "EANM/ESC Procedural Guidelines for Myocardial Perfusion Imaging in Nuclear Cardiology," Eur. J. Nucl. Med. Mol. Imaging 32, 855 (2005).
[3] A. Fathala, "Myocardial Perfusion Scintigraphy: Techniques, Interpretation, Indications and Reporting," Ann. Saudi Med. 31, 625 (2011).
[4] A. Rios, "Nuclear Medicine Imaging," Physics 241, Stanford University, Winter 2017.
[5] E. Pinilla, "Nuclear Medicine: Positron Emission Tomography," Physics 241, Stanford University, Winter 2018.
[6] G. Klaris, "The Future of Nuclear Medicine," Physics 241, Stanford University, Winter 2016.