 |
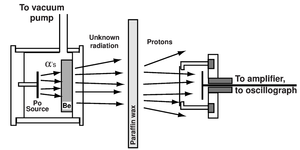
| Fig. 1: A schematic diagram of the experiment James Chadwick used to discover the neutron in 1932. (Source: Wikimedia Commons) |
James Chadwick was born in Cheshire, England, on 20th October, 1891. He graduated from Manchester University in 1908 and went on to graduate from the Honours School of Physics in 1911. After graduation he spent two years working in Physical Laboratory in Manchester, where he worked on various radioactivity problems, gaining his M.Sc. degree in 1913. After being interned in the Zivilgefangenenlager, Ruhleben during World War I, Chadwick returned to England to continue his research. Chadwick continued to move up the ladder in the world of science when he was elected Fellow of Gonville and Caius College (1921-1935) and became Assistant Director of Research in the Cavendish Laboratory (1923). In 1927 he was elected a Fellow of the Royal Society. [1]
In 1932, Chadwick made a fundamental discovery in the domain of nuclear science. Chadwick was fascinated by an experiment done by Frdric and Irne Joliot-Curie that studied the then-unidentified radiation from beryllium as it hit a paraffin wax target. The Curies found that this radiation knocked loose protons from hydrogen atoms in that target, and those protons recoiled with very high velocity. In 1932, Chadwick tried similar experiments himself and hypothesized that the radiation ejected by the beryllium was, in fact, a neutral particle with approximately the same mass as a proton. Fig. 1 depicts a schematic diagram of the experiment done by Chadwick, following on experiments done by the Curies. He later tried other targets including helium, nitrogen, and lithium, which led him to determine that the mass of the new particle was in fact just slightly greater than the mass of the proton. [1] This is reflected in the current understanding of the mass of a neutron as 1.008701 amu or 1.6750 × 10-24 g and the mass of a proton as 1.007316 amu or 1.6727 × 10-24 g. [2]
After only about two weeks of experimentation, Chadwick wrote a paper in which he proposed that the evidence favored the neutron rather than the gamma ray photons as the correct interpretation of the radiation. Only a few months later, in May 1932, Chadwick submitted a paper announcing the discovery of the Neutron. The existence of a neutron as a new fundamental particle was firmly established by 1934. Chadwick was awarded the Nobel Prize in 1935 for its discovery. [3]
Chadwick's discovery of the neutron was the final piece in understanding the atomic puzzle and sparked a revolution leading to the nuclear age and the creation of nuclear weapons. [4]
© Maya Kuppermann. The author warrants that the work is the author's own and that Stanford University provided no input other than typesetting and referencing guidelines. The author grants permission to copy, distribute and display this work in unaltered form, with attribution to the author, for noncommercial purposes only. All other rights, including commercial rights, are reserved to the author.
[1] A. Brown, The Neutron and the Bomb: A Biography of Sir James Chadwick (Oxford University Press, 1997).
[2] D. W. Oxtoby and H. P. Gillis, Principles of Modern Chemistry, 5th Ed. (Brooks Cole, 2002).
[3] M. Oliphant, "The Beginning: Chadwick and the Neutron," Bull. Atom. Sci. 38, 14 (1982).
[4] K. Fischer, A Brief History of Pulsed Neutron Generation," Physics 241, Stanford University, Winter 2015.