 |
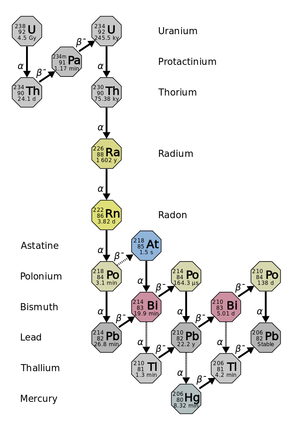
| Fig. 1: Radioactive decay chains of uranium. (Source: Wikimedia Commons) |
Radon, atomic number 86 on the periodic table, is a colorless, odorless, and tasteless gas. Formed during the sequential radioactive decay of uranium (see Fig. 1), Radon has a very short half-life of 3.8 days. Radon's radioactive decay to Po-218 releases an α particle. α particles can damage human cells, and, with sufficient exposure and latency, can cause cancer.
Given Radon's short half-life, one may suspect that an individual's exposure to it gas would be exceedingly small. Uranium, however, is one of the most common radioactive elements on Earth and has a very long half-life (4.5 × 109 years). Uranium decay countinually replentishes the Radon supply, so the amount of Radon in the environment is constant on the scale of a billion years.
Once generated, the Radon gas diffused through fissures and cracks in the soil and out into the atmosphere. In the United States, outdoor air levels of Radon are generally around 0.4 picocuries per liter (pCi/L). [1] The Radon seeping through the soil can also come into contact with building foundations. Cracks in foundations can give Radon the ability to seep inside the structure. Indoor Radon air levels in the United States average about 1.3 pCi/L; four times the outside level. [1] Once inside a structure, due to its density (atomic number of 86 and an atomic mass of 222), Radon will sink to low lying areas such as basements, crawl spaces, and sump-pump pits. As a result, Radon levels in residential basements are higher than the average indoor levels. It is important to note, however, that uranium deposits are not evenly distributed around the globe. As a result, Radon levels vary country-to-country, region-to-region, neighborhood-to-neighborhood, and even house-to-house and room-to-room.
In addition to the air and soil, water can also have Radon contamination. However, for public drinking water systems, Radon levels are low because the water treatment process involves aerosolizing the water, which releases the dissolved Radon to the atmosphere. However, privately owned well water is a different story, and could represent a significant source of Radon exposure. While ingestion is a potential risk, most of the Radon exposure from well water comes from inhalation of Radon released into the air during showering and other household activities. [1]
As Radon decays, the resulting atoms are known as Radon progeny or daughters. To have a health effect, Radon, or its progeny, must gain access to the body. Radon, as a gas, enters the body by inhalation. The progeny, as solids with an electrostatic charge, adhere to very small dust particles. These small dust particles can then carry the progeny into the lung where they attach to the lung epithelium. [2]
As Radon decays it emits α-radiation. As the Radon progeny decay, they emit either α or β radiation (Fig. 1). α and β radiation can cause mutations in the lung epithelial cells' DNA. Ionizing radiation, including α radiation, is not particularly efficient at producing point mutations, but it does produce large interstitial deletions and reciprocal translocations. This is most likely the first of many steps by which alpha particles can induce cancer. [2] Without a cellular DNA repair mechanism, and with significant exposure and latency periods, the mutation can become a lung cancer. Given that alpha radiation can only travel very short distances in the body, Radon's health effect is primarily localized to the lung. [2]
The association between Radon exposure and lung cancer was first recognized among underground miners more than 100 years ago. [3] Subsequent epidemiological studies of underground miners who are heavily exposed to Radon, have shown a very strong and consistent association between Radon exposure and lung cancer. This association has been found in multiple studies which demonstrated a linear dose-response relationship (e.g., the higher the Radon exposure, the greater risk of lung cancer). [2] Extrapolating the risk assessment from the miners to the effect of Radon exposure in the general population is difficult because the miners had much higher levels of Radon exposure. However, in 1999 the National Academy of Sciences published their extensive review of the subject concluding that there are between 15,400 to 21,800 Radon-induced lung cancer deaths occurring each year in the United States. [2]
The Environmental Protection Agency (EPA) recommends testing homes for Radon with both short-term and long-term tests. However, only small fractions of homes are ever tested. If the long term test is 4 pCi/L or more, the EPA recommends remediation. [1] The EPA estimates over 5% of homes in the United States have elevated Radon levels. [1] There are several effective strategies to reduce Radon from the home. The most common system is a vent pipe system that pulls Radon from beneath the house and vents it to the outside. For most homes, this can be accomplished at about the same costs as other common home repairs.
Despite known Radon contamination of the indoor environment, thousands of Radon-caused lung cancer deaths occurring each year, and relatively low costs to remediate, new homes are built without Radon mitigation systems, and existing homes are not being tested for Radon contamination. Currently, the EPA is proposing to eliminate funds to promote Radon awareness and oversee profession testing. [4] Therefore, States should consider legislation regarding building/real estate codes to ensure Radon testing is performed with each home sale, and remediation for levels exceeding 4 pCi/L.
© Hannah Hales. The author warrants that the work is the author's own and that Stanford University provided no input other than typesetting and referencing guidelines. The author grants permission to copy, distribute and display this work in unaltered form, with attribution to the author, for noncommercial purposes only. All other rights, including commercial rights, are reserved to the author.
[1] "A Citizen's Guide to Radon" U.S. Environmental Protection Agency, EPA 402/K-12/002, May 2012, p. 7
[2] Health Effects of Exposure to Radon: BEIR VI (National Academies Press, 1999), pp. 1-3, 38.
[3] J. M. Samet, "Indoor Radon and Lung Cancer: Estimating the Risks," Western J. Med. 156, 25 (1992)
[4] M. Martin, "Budget Cuts Could Lead to More Radon Deaths Newsweek," Newsweek, 30 Apr 17.