 |
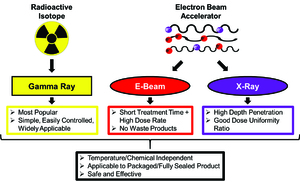
| Fig. 1: Comparison of radiation sources for radiation sterilization. [1-7] (Source: E. Goronzy) |
Commercial radiation sterilization has existed since the late 1950s and has grown tremendously in popularity over the last 60 years. [1] Radiation sterilization relies on ionizing radiation, primarily gamma, X-ray or electron radiation, to deactivate microorganisms such as bacteria, fungi, viruses and spores. Due to numerous advantages over heat or chemical based sterilization techniques, this method is particularly attractive in medicine and healthcare-related fields. [2] For example, radiation sterilization is readily applied during tissue allograft preparation, pharmaceutical packaging and medical device manufacturing. [3]
Radiation can be lethal to biological organisms by inducing genetic damage and chemical changes in key biological macromolecules. [3] During sterilization treatment, the sample of interest is bombarded with high energy electrons or high energy electromagnetic radiation, which leads to the formation of extremely unstable free radicals, molecular ions and secondary electrons. These radiation products then react with nearby molecules to fracture and alter chemical bonds. DNA in particular is highly sensitive to the damaging effects of radiation and will break, depolymerize, mutate and alter structure upon exposure to ionizing radiation. Incomplete repair of DNA damage ultimately leads to loss of genetic information and cell death. Thus, radiation can kill harmful microorganisms and be used as a sterilization technique.
The sensitivity of a given biological organism to radiation is given by the decimal reduction dose (D10 value), the dose of radiation which leads to a 10-fold reduction in microorganism population. [3] In order to be effective, sterilization treatment must be dosed to account for the D10 values of microorganisms present, the initial level of bioburden, and the diversity of the bioburden in the sample. Furthermore, the optimal radiation dose depends on the desired sterility assurance level (SAL), the probability that a microorganism will survive the sterilization procedure. A SAL of 10-6 or better is usually desired. For example, a standard radiation dose of 25 kGy used to treat a product contaminated with Salmonella typhimurim (D10 = 0.3 kGy) or Vibrio cholerae (D10 = 0.48 kGy) would result in a SAL less than 10-24; the same dose applied to a sample contaminated with HIV (D10= 4.0 - 8.4 kGy) would only result in a SAL of ~10-3 - 10-6. Overall, radiation dosage is not absolute and must be optimized given the properties of the sample and the level of sterility required.
Three forms of radiation commonly used for commercial radiation sterilization include gamma radiation, electron beam (e-beam) radiation and X-ray radiation (Fig. 1). [1]
Gamma radiation sterilization is the most popular form of radiation sterilization. [1,4] Co-60 and, to a lesser extent, Cs-137 serve as radiation sources and undergo decomposition to release high energy gamma rays. The produced electromagnetic radiation is highly penetrating and can kill contaminating microorganisms. Both radioisotopes are viable sources of radiation due to their highly stability (with half-lives >5 years) and gamma emission properties. However, Co-60 tends to be favored because it can be easily manufactured from natural metal, is not fissile or flammable and is less soluble in water.
Radioactive Co is formed inside a nuclear reactor by neutron bombardment of the abundant, non-radioactive Co-59 isotope. [1,4] Co-60 atoms then decay to nonradioactive Ni-60 atoms by emitting one electron and two gamma rays at energies of 1.17 MeV and 1.33 MeV. Gamma rays are released in an isotropic fashion and are not high enough in energy to induce radioactivity in other materials. In general, production requirements for Co-60 makes gamma sterilization at individual hospital sites unattractive but it is feasible through large scale commercial manufacturing. Gamma based radiation sterilization has been deemed safe and effective by a number of government and public health agencies including the US Center for Disease Control and Prevention, the Food and Agriculture Organization, the United Nations and the World Health Organization.
Sterilization can alternatively be accomplished using electron beam irradiation. [2] High energy electrons capable of inducing biological damage are generated by electron beam accelerators. In most cases, electron energies of ~10 MeV are used, but the exact energies can be tuned to optimize penetration depth and limit breakdown of the irradiated material.
Gamma irradiation and e-beam irradiation differ in sample penetration depth, exposure time required for effective sterilization and product compatibility. [2,3] Because the penetration ability of electrons is lower than that of gamma rays, e-beam sterilization is limited in application to lower density or smaller products. However, e-beam sterilization can use higher dosages and shorter treatment times (seconds vs. min/hours) as compared to gamma radiation sterilization, allowing for higher throughput and reducing negative effects on treated products. In terms of cost, e-beam sterilization is equivalent to or less expensive than gamma sterilization. [4]
Electron beam accelerators will also generate X-rays for sterilization. [1,4] X-rays are produced when high energy electrons from the accelerator interact with high atomic number nuclei, such as atoms of tungsten or tantalum. In a process known as Bremsstrahlung, the deceleration of the electron when passing the nucleus results in the release of X-rays. Electron energies of 5-7 MeV are commercially used; the energies of the resultant X-rays lie along a spectrum ranging from zero to the energy of the electron beam.
In practice, X-rays used for sterilization can be more penetrating than either gamma-rays or electron beams. [4] They are largely directional since generated X-rays propagate in the same direction as the incident electron. Thus, a concerted stream of X-rays is sent towards the product of interest and multiple rows of products can be sterilized simultaneously. Of radiation sterilization techniques, X-ray sterilization can achieve the highest dose uniformity ratio (DUR), the ratio between maximum and minimum dose required for sterilization. [5-7] DUR measures the range of doses delivered to the product and is important to optimize for irradiation sensitive materials in order to minimize degradation.
Radiation based sterilization methods offer numerous advantages over traditional chemical or heat-based sterilization: [1-4]
Terminal Processing: Due to the penetration depth of ionizing radiation, products can be processed in their fully sealed, final packaging. This limits risk of contamination following sterilization.
Cold Method/Temperature Independence: Temperature increases during treatment are minimal. Furthermore, radiation sterilization has no heat dependence and is efficient at both ambient temperature and sub-zero temperatures. It is compatible with temperature sensitive materials, such as pharmaceuticals and biological samples.
Chemical Independence: No volatile or toxic chemicals are needed. In the case of X-ray or e-beam irradiation, no end products requiring disposal are generated during the procedure.
No residue: Radiation leaves no residue on the sterilized product.
Flexibility: Radiation can sterilize products of any phase (gaseous, liquid or solid materials), products with variable density, size or thickness, and homogeneous or heterogeneous systems. Furthermore, sterilization can be conducted at any temperature and any pressure.
Time efficiency: E-beam sterilization can be completed within seconds to minutes.
Sterility assurance level (SAL): Radiation treatment can yield a high SAL of 10-6 or better, ensuring that less than one out of a million microorganisms survive the sterilization procedure.
However, radiation sterilization techniques do have a number of drawbacks: [2-4]
Instrumentation: Capital costs are high and specialized facilities are often needed. Gamma radiation requires a nuclear reactor; E-beam/X-ray radiation are generated using electron beam accelerators.
Product Degradation: Radiation based methods are not compatible with all materials and can cause breakdown of the packaging material and/or product. Common plastics such as polyvinyl chloride (PVC), acetal and polytetrafluoroethylene (PTFE) are sensitive to gamma radiation. The high energies involved in e-beam radiation can also lead to main chain scission (breaking of the long chain backbone) and chemical crosslinking of packaging polymers.
Radioactive material: When gamma radiation is used as an ionization source, radiation sterilization requires handling and disposal of radioactive material. Note that, at commonly used radiation levels, irradiation with gamma rays does not induce radioactivity in the treated sample itself.
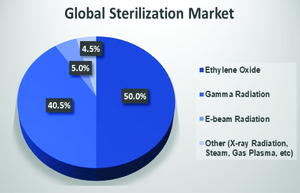 |
| Fig. 2: Global Sterilization Market Volume Shares, by Sterilization Technology. [4] (Source: E. Goronzy) |
The value of the global sterilization market was $4.69 billion in 2016 and is estimated to increase to $6.93 billion by 2021. [4] Gamma sterilization is the most prevalent form of radiation-based sterilization, but the increasing availability of high power electron accelerators has made e-beam and X-ray-based technologies popular alternatives. The sterilization market is comprised of about 40.5% gamma-based sterilization, 4.5% e-beam sterilization, 50% ethylene oxide sterilization (a gaseous sterilization method) and 5% other, such as x-ray sterilization (Fig. 2).
In 2017, over 200 gamma irradiation facilities could be found spread throughout 50 countries. [4] These facilities utilize approximately 400 million curies of Co-60 each year to sterilize 400 million cubic feet of product. Electron beam sterilization facilities were somewhat less common, with fewer than 75 healthcare sterilization facilities estimated operational worldwide. X-ray sterilization is only a relatively recent development, propelled in growth by technological developments in high powered electron accelerators. [1] The first commercial facilities for X-ray sterilization of medical products appeared in 2010 and fewer than 5 were present in 2017. [4]
Currently, an estimated 40-50% of all disposable medical products are sterilized using radiation. [8] For example, sutures, surgical gloves, gowns, face masks, syringes, sticking plasters, dressings and other single use healthcare supplies are all processed using gamma radiation.[2,3,9] Radiation sterilization is particularly popular for such products since it can be applied to the fully packaged, sealed item, thus limiting the potential for recontamination by eliminating post-sterilization packaging steps. The compatibility of medical device components and packaging materials with radiation requires consideration. While many materials, such as metals, are unaffected by sterilization levels of radiation, polymers can undergo chemical reactions when irradiated, leading to changes in physical properties. Dose optimization, the use of less radiation-sensitive polymers and the inclusion of polymer stabilizers in packaging and device materials can limit undesired side reactions during gamma sterilization.
Standards for radiation sterilization of medical products are set by the International Organization for Standardization (ISO). [10,11] In 1995, the ISO released the first edition of ISO-11137 outlining validation methods, quality controls and requirements for all activities related to the sterilization processes. The United States played a considerable role in shaping the document and participated through the Association for Advancement of Medical Instrumentation. The final name adopted in the US was ANSI/AAMI/ISO 11137. The document contains three parts: "Part 1: Requirements for development, validation and routine control of a sterilization process for medical devices", "Part 2: Establishing the sterilization dose", and "Part 3: Guidance on dosimetric aspects". ISO 11137 has been amended and updated multiple times to incorporate the latest standards.
Gamma irradiation is a common method for the sterilization of connective tissue allografts, such as skin, cartilage, bone, tendons, heart values and corneas. [11] A key concern for tissue allografts is risk of disease transmission to the recipient. Hazardous microorganisms may be of donor origin or may have been introduced during tissue procurement, processing, storage and transfer. To reduce the possibility of bacterial, fungal or viral disease transmission, tissue samples must be sterilized before introduction into the recipient. Gamma irradiation allows for targeted reduction of microbials and viruses, often without drastic alterations of tissue properties. However, radiation will induce some physical and chemical changes in the graft. A compromise is required to ensure high enough radiation for microbial deactivation but low enough radiation to maintain the properties of the allograft. A dose of 25 kGy of radiation was long considered the gold standard for tissue treatment and has been shown sufficient to inactivate many bacteria, fungi, and enveloped and non-enveloped RNA and DNA viruses. [3,11] However, tissue banks often modify their dosage from the reference 25 kGy and use doses between 15 - 35 kGy in order to match the bioburden of the sample, achieve the desired SAL and maintain tissue integrity. The ISO provides guidelines for dose validation to help tissue banks determine the optimal radiation dose. Research is currently underway to determine the exact effects of radiation treatment on the biological quality and clinical efficacy of the allograft.
© Isabel Goronzy. The author warrants that the work is the author's own and that Stanford University provided no input other than typesetting and referencing guidelines. The author grants permission to copy, distribute and display this work in unaltered form, with attribution to the author, for noncommercial purposes only. All other rights, including commercial rights, are reserved to the author.
[1] B. P. Fairand, Radiation Sterilization for Health Care Products: X-Ray, Gamma, and Electron Beam (CRC Press, 2001).
[2] M. Silindir and A. Y. Özer, "Sterilization Methods and the Comparison of E- Beam Sterilization with Gamma Radiation Sterilization," FABAD J. Pharm. Sci. 34, 43 (2009).
[3] F. Adrovic, ed., Gamma Radiation (InTech, 2012).
[4] "A Comparison of Gamma, E-beam, X-ray and Ethylene Oxide Technology for the Industrial Sterilization of Medical Devices and Healthcare Products," Gamma Industry Processing Alliance, 31 Aug 17.
[5] W. Rogers, Healthcare Sterilization: Challenging Practices Volume 2 (Smithers Information Itd., 2014).
[6] B. Ratner et al., ed., Biomaterial Science: An Introduction to Materials in Medicine (Elsevier, 2013).
[7] Y. Qin, ed., Medical Textile Materials (Elsevier, 2016).
[8] J. Masefield, "Reflections on the evolution and current status of the radiation industry," Radiat. Phys. Chem. 71 (2004).
[9] D. Dempsey, R. Ramachandran and M. Thirucote, "Sterilization of Medical Devices: A Review," J. Biomater Appl. 3, 3 (1989).
[10] A. Booth, ed., Sterilization of Medical Devices (Interpharm Press, 1999).
[11] R. Singh, D. Singh and A. Singh, "Radiation Sterilization of Tissue Allografts: A Review," World J. Radiol. 8, 365 (2016).