 |
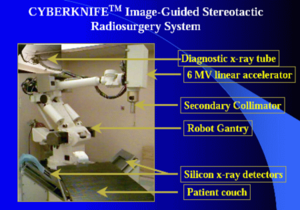
| Fig. 1: Main schematic of a CyberKnife system. (Source: Wikimedia Commons) |
In radiation therapy, high-energy radiation is directed at tumors and cancer cells. This report will focus on external-beam radiation: radiation in the form of x-ray or gamma that is delivered from a machine outside of the body. Radiation works by damaging the DNA of cells, resulting in cell death. [1] Radiation induced death is not specific to cancer cells, which is why many systems seek to minimize the area that is exposed to radiation.
Radiation therapy systems seek to maximize elimination of cancer cells and minimize side effects. One serious side effect is secondary cancer caused by affected non-cancerous cells. [2] Because it is not possible to identify and distinguish single cancer cells, modern therapy systems seek to maximize their therapeutic ratio.
Stereotactic radiation therapy systems accomplish this by immobolizing the target tumor, accounting for tumor motion, minimizing radiation to the tumor, and delivering a high dosage to this small volume of mass. This dosage is around 24-48 Gy. A Gy, or gray, is a unit of ionizing radiation dose, defined as the absorption of one joule of radiation per kilogram of target area. [3,4] Treatment protocols also use computed tomography scans, magnetic resonance imagery, and positron emission tomography to identify tumors. Advances in these imaging systems may increase our ability to distinguish cancer cells from normal cells. [5]
There are many stereotactic radiation therapy systems that are in use today. One example is the CyberKnife system developed at Stanford (Fig. 1). What distinguishes CyberKnife from other stereotactic systems is that it uses a linear accelerator - 6 MV LINAC - on the end of a robotic arm to deliver radiation to the target while maintaining accuracy. Other systems such as Gamma Knife have a mechanical accuracy of 0.2 mm, and studies have found CyberKnife to be comparable. [6] CyberKnife's distinguishing feature, the movable arm, allows for fast re-positioning of the radiation source. Tracking systems have been developed to compensate for respiratory motion during radiation, minimizing the damage to non-cancerous cells caused by these cells moving into the target during motion. This works by enabling the robotic arm to follow the tumor as it moves. With fixed arm systems, the beam would remain in place as the tumor moved out of the frame and surrounding tissue moved in, causing unnecessary damage to healthy tissue. [7] Along with the advantages, a CyberKnife is a sizable investment at around $3-5 million per machine. [8]
© Cody Carlton. The author warrants that the work is the author's own and that Stanford University provided no input other than typesetting and referencing guidelines. The author grants permission to copy, distribute and display this work in unaltered form, with attribution to the author, for noncommercial purposes only. All other rights, including commercial rights, are reserved to the author.
[1] D. Le, "Radiation Therapy for Cancer," Physics 241, Stanford University, Winter 2017.
[2] S. Ali, "Radiation Oncology: Mechanism and Resistance," Physics 241, Stanford University, Winter 2012.
[3] L. M. Halasz and J. K. Rockhill, "Stereotactic Radiosurgery and Stereotactic Radiotherapy for Brain Metastases," Surg. Neurol. Int. 4, Suppl. 4, S185 (2013).
[4] E. Göbel, I. M. Mills, and A. J. Wallard, "The International Systems of Units (SI)," Organisation Intergouvernementale de la Convention du Mètre, March 2006.
[5] S. Noda et. al., "Technological Advances in Radiation Oncology for Central Nervous System Tumors," Semin. Radiat. Oncol. 19, 179 (2009).
[6] W. Hara, S. G. Soltys, and I. C. Gibbs, "CyberKnife Robotic Radiosurgery System for Tumor Treatment," Expert Rev. Antican. 7, 1507 (2007).
[7] A. Schweikard et. al., "Robotic Motion Compensation for Respiratory Movement During Radiosurgery," Comput. Aided Surg. 5, 263 (200).
[8] S. Saul, "Geography Has a Role in Medicare Cancer Coverage," New York Times, 16 Dec 08.