 |
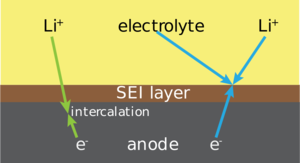
| Fig. 1: SEI formation in Li-ion battery systems. (Source: Wikimedia Commons) |
Increasing demand for high-density energy storage is driving a search for new alternatives to the high cost and low availability of the lithium ion batteries used on a large proportion in today's time. Sodium-ion batteries offer an affordable and earth-abundant alternative to lithium-ion batteries. [1]
While significant research progress has been made in developing electrode materials with high charge storage capacities and rate capability, the success of sodium-ion batteries is hindered by unstable formation of the surface-electrolyte interphase (SEI). [2]
While SEI formation has been the focus of significant research interest in lithium-ion systems, it is still poorly understood in the context of sodium-ion batteries; due to notable differences between sodium and lithium, including increased sodium reactivity and increased solubility of degradation products, which means that knowledge does not directly translate from lithium research into sodium. This is the reason why it is essential to understand it further. Fig. 1 shows the researched SEI layer formation in lithium ion systems. [1]
The solid electrolyte interphase (SEI) is a layer that forms at the anode surface for all alkali metal ion batteries which utilize liquid electrolytes. [3]
Within sodium ion batteries, sodium ions are transferred from a cathode to an anode through an electrolyte during charging and then from the anode to the cathode during discharging. As the battery is cycled, a solid layer builds up at the interface between the electrolyte and the electrodes. This layer is called the solid electrolyte interphase (SEI) and it is formed mainly from decomposition products of the electrolyte. [2,3]
The SEI is of crucial importance to the battery as it protects the anode by inhibiting the transfer of electrons from the anode to the electrolyte, whilst also allowing sodium ions to transfer from the electrolyte to the anode. The ideal SEI is therefore both an ionic conductor and an electrical insulator. [3]
Hence it is proving to be an essential component in battery systems can be a potential asset towards improving conductivity of ions especially at the electrodes. However the formation of the SEI results in an irreversible capacity loss, so it is essential to understand it, to make the optimum use of it. [3]
 |
| Fig. 2: Sodium Ion Battery System. (Source: Wikimedia Commons) |
The operation of Na-ion batteries is similar to that of Li-ion batteries, and it commonly uses sodium hexafluorophosphate (NaPF6) dissolved in a mixture of alkyl carbonates as the electrolyte solution and two high-/low-potential operation electrodes exhibiting reversible redox reactions with Na ions. This is illustrated in Fig. 2. [4]
Due to the limitations posed by Na-ion intercalation into graphite (e.g., Na-ion intercalation leads to a low storage capacity of 248 mA h/g versus < 372 mAh/g for Li-ions), hard carbons are used as the intercalation material for the anode in Na-ion batteries. The reactions that take place in Na-ion batteries lead to the formation of a nonuniform SEI layer that consists of inorganic compounds, sodium carbonate and NaCO3R (R = alkyl) in ethylene-carbonate-/propylene-carbonate-based (EC-/PC-based) electrolytes.
However, the reaction of the hard carbon is reversible, as the sodium cell is composed of inorganic compounds rather than the organic compounds one has in Li-ion battery systems - that cause irreversibility. [4,5]
For high capacity anodes in sodium ion batteries, it is found that continued cycling of the battery ruptures the SEI and exposes new areas of the anode surface where the electrolyte will reduce. This effect is prominent as it is hampered by the higher solubility of SEI components such as sodium salts in comparison to the lithium analogues. The SEI layer can lead to loss of charge carriers and slower ionic transport among other factors that contribute to battery failure. [4]
Recent work has suggested that the initially formed SEI layer can be tuned to improve the ionic transport within the SEI layer of Li-ion and Na-ion batteries. It is possible to design a stable SEI layer by pre-cycling the electrode in desired Na-based electrolyte and that the ionic transport can be tuned by preforming SEI layers of specific compositions. [4,6]
However, the design of such preformed layers demands a better knowledge about the mechanisms of ion migration through them. Thus, having a deeper understanding of the SEI layer stability and ionic transport through SEI components in NaBs is a major step in achieving the stability of the SEI layer, ultimately improving the capacity retention of the batteries. Such understanding requires a detailed characterisation of the kinetics of ionic transport, which is usually characterised via evaluation of mechanistic pathways and activation barriers via first- principles analyses. [5,6]
For the migration of ions through SEI blocks, different neutral defects in the SEI compounds were investigated along various ionic diffusion channels. Several pathways such as vacancy diffusion, direct hopping, knock-off, and concerted-exchange mechanisms were characterised, although the high formation energies of some defects would not allow ionic migration. The calculation of the defect formation and migration energies leads to the conclusion that some defects could be easy to form, but migrate very slowly and vice versa. [4]
A further finding has been the understanding of how the high reactivity of sodium metal causes electrolyte degradation and SEI formation occur at the sodium metal surface even without electrochemical cycling; which makes it even more concerning during the manufacture of the batteries. There seems to be a potential research being undergone by using the the influence of fluoroethylene carbonate (FEC) electrolyte additive to mitigate effects of self discharge in this maner. [5]
New battery technologies are developing, especially with the aim to develop a practical, safe, smaller and lower-cost batteries. It has led to the development of sodium-air rechargeable batteries it is necessary to eliminate the formation of dendritic deposits (on charge), and to increase the power capability of the batteries. [6]
Research has shown that it is beneficial to replace the metallic-lithium anode by liquid sodium and to operate the sodiumoxygen cell above the sodium melting point (97.8°C). One of the main conclusions of the research was that in order to create a protective SEI on alkali-metal anodes, it is essential that the equivalent volumes of the SEI materials be greater than that of the metal anode. Only in this way does the SEI completely cover the anode surface and stop corrosion. If not, the anode will continue to corrode. Since the solid electrolyte interphase, formed on the anode during battery operation is the most vulnerable factor in the safety and cycle life of the sodium-air battery, the formation of a proper SEI is a critical process. [6]
Further research indicates however, that upon comparing the SEI formed in Na-air versus Li-air batteries, the charge-transfer resistance associated with the SEU is significantly higher in the case of the Na-anode cell than for the Li-anode cell. In the sodium-based system, that extends over a high 30 Ω.cm2; growing even further with time and developing into a different shape. This indicates that the interface between hard carbon and the electrolyte becomes more resistive with time, and its nature is more complex. It is hence essential to understand, this type of system; especially for longer term applications, which would be essential. [3,6]
With developing technologies, it is essential to understand these types of systems, especially the impacts of an essential component like the SEI, otherwise it might always prove to be a limitation in progression for the future.
© Valmik Lakhlani. The author warrants that the work is the author's own and that Stanford University provided no input other than typesetting and referencing guidelines. The author grants permission to copy, distribute and display this work in unaltered form, with attribution to the author, for noncommercial purposes only. All other rights, including commercial rights, are reserved to the author.
[1] F.A. Soto et al., "Understanding Ionic Diffusion through SEI Components for Lithium-Ion and Sodium-Ion Batteries: Insights from First-Principles Calculations" Chem. Mater. 30, 3315 (2018).
[2] J.M. Martinez et al., "Effect of the Electrolyte Composition on SEI Reactions at Si Anodes of Li-Ion Batteries." J.Phys. Chem.C 13, 7060 (2015).
[3] C. Bommier and X. Ji, "Electrolytes, SEI Formation, and Binders: A Review of Non-electrode Factors for Sodium-Ion Battery Anodes," Small 14, 1703576 (2018).
[4] R. Mogensen, D. Brandell and R. Younesi, "Solubility of the Solid Electrolyte Interphase (SEI) in Sodium Ion Batteries" ACS Energy Lett. 1, 1173 (2016).
[5] N. Weadock et al., "Determination of Mechanical Properties of the SEI in Sodium Ion Batteries Vvia Colloidal Probe Mmicroscopy" Nano Energy 2, 2056 (2018).
[6] E. Peled and S. Menkin, "Review-SEI - Past, Present and Future," J. Electrochem. Soc. 164, A1073 (2018).