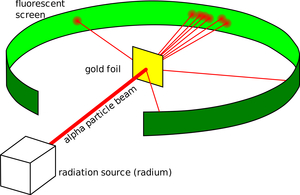
 |
| Fig. 1: A three-dimensional view of an apparatus similar to Geiger and Marsden's final cylindrical iteration, clearly showing the scattering of alpha particles by gold foil. (Source: Wikimedia Commons) |
The Rutherford Gold Foil Experiment offered the first experimental evidence that led to the discovery of the nucleus of the atom as a small, dense, and positively charged atomic core. Also known as the Geiger-Marsden Experiments, the discovery actually involved a series of experiments performed by Hans Geiger and Ernest Marsden under Ernest Rutherford. With Geiger and Marsden's experimental evidence, Rutherford deduced a model of the atom, discovering the atomic nucleus. His "Rutherford Model", outlining a tiny positively charged atomic center surrounded by orbiting electrons, was a pivotal scientific discovery revealing the structure of the atoms that comprise all the matter in the universe.
The experimental evidence behind the discovery involved the scattering of a particle beam after passing through a thin gold foil obstruction. The particles used for the experiment - alpha particles - are positive, dense, and can be emitted by a radioactive source. Ernest Rutherford discovered the alpha particle as a positive radioactive emission in 1899, and deduced its charge and mass properties in 1913 by analyzing the charge it induced in the air around it. [1] As these alpha particles have a significant positive charge, any significant potential interference would have to be caused by a large concentration of electrostatic force somewhere in the structure of the atom. [2]
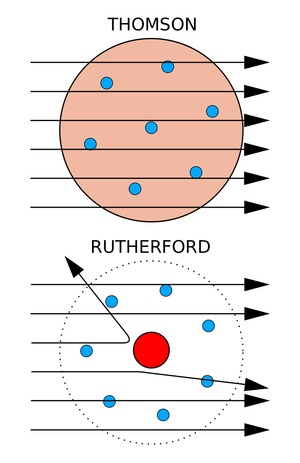 |
| Fig. 2: A comparison between J.J. Thompson's "plum pudding" atomic model and the Rutherford model and its nucleus. Alpha particles and their scattering or lack thereof are depicted by the paths of the black arrows. (Source: Wikimedia Commons) |
The scattering of an alpha particle beam should have been impossible according to the accepted model of the atom at the time. This model, outlined by Lord Kelvin and expanded upon by J. J. Thompson following his discovery of the electron, held that atoms were comprised of a sphere of positive electric charge dotted by the presence of negatively charged electrons. [3] Describing an atomic model similar to "plum pudding," it was assumed that electrons were distributed throughout this positive charge field, like plums distributed in the dessert. However, this plum pudding model lacked the presence of any significant concentration of electromagnetic force that could tangibly affect any alpha particles passing through atoms. As such, alpha particles should show no signs of scattering when passing through thin matter. [4] (see Fig. 2)
Testing this accepted theory, Hans Geiger and Ernest Marsden discovered that atoms indeed scattered alpha particles, a experimental result completely contrary to Thompson's model of the atom. In 1908, the first paper of the series of experiments was published, outlining the apparatus used to determine this scattering and the scattering results at small angles. Geiger constructed a two meter long glass tube, capped off on one end by radium source of alpha particles and on the other end by a phosphorescent screen that emitted light when hit by a particle. (see Fig. 3) Alpha particles traveled down the length of the tube, through a slit in the middle and hit the screen detector, producing scintillations of light that marked their point of incidence. Geiger noted that "in a good vacuum, hardly and scintillations were observed outside of the geometric image of the slit, "while when the slit was covered by gold leaf, the area of the observed scintillations was much broader and "the difference in distribution could be noted with the naked eye." [5]
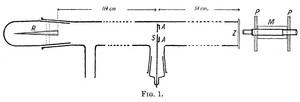 |
| Fig. 3: The schematics for the original two meter long tube that Geiger constructed and used to first detect the scattering of alpha particles by the atomic nucleus. At the point labeled R is the radon particle emission source, and Z the detector screen. (Source: Wikimedia Commons) |
On Rutherford's request, Geiger and Marsden continued to test for scattering at larger angles and under different experimental parameters, collecting the data that enabled Rutherford to further his own conclusions about the nature of the nucleus. By 1909, Geiger and Marsden showed the reflection of alpha particles at angles greater than 90 degrees by angling the alpha particle source towards a foil sheet reflector that then would theoretically reflect incident particles at the detection screen. Separating the particle source and the detector screen by a lead barrier to reduce stray emission, they noted that 1 in every 8000 alpha particles indeed reflected at the obtuse angles required by the reflection of metal sheet and onto the screen on the other side. [6] Moreover, in 1910, Geiger improved the design of his first vacuum tube experiment, making it easier to measure deflection distance, vary foil types and thicknesses, and adjust the alpha particle stream' velocity with mica and aluminum obstructions. Here he discovered that both thicker foil and foils made of elements of increased atomic weight resulted in an increased most probable scattering angle. Additionally, he confirmed that the probability for an angle of reflection greater than 90 degrees was "vanishingly small" and noted that increased particle velocity decreased the most probably scattering angle. [7]
Backed by this experimental evidence, Rutherford outlined his model of the atom's structure, reasoning that as atoms clearly scattered incident alpha particles, the structure contained a much larger electrostatic force than earlier anticipated; as large angle scattering was a rare occurrence, the electrostatic charge source was only contained within a fraction of the total volume of the atom. As he concludes this reasoning with the "simplest explanation" in his 1911 paper, the "atom contains a central charge distributed through a very small volume" and "the large single deflexions are due to the central charge as a whole." In fact, he mathematically modeled the scattering patterns predicted by this model with this small central "nucleus" to be a point charge. Geiger and Marsden later experimentally verified each of the relationships predicted in Rutherford's mathematical model with techniques and scattering apparatuses that improved upon their prior work, confirming Rutherford's atomic structure. [4, 8, 9] (see Fig. 1)
With the experimentally analyzed nature of deflection of alpha rays by thin gold foil, the truth outlining the structure of the atom falls into place. Though later slightly corrected by Quantum Mechanics effects, the understanding of the structure of the the atom today almost entirely follows form Rutherford's conclusions on the Geiger and Marsden experiments. This landmark discovery fundamentally furthered all fields of science, forever changing mankind's understanding of the world around us.
© George Sivulka. The author grants permission to copy, distribute and display this work in unaltered form, with attribution to the author, for noncommercial purposes only. All other rights, including commercial rights, are reserved to the author.
[1] E. Rutherford, "Uranium Radiation and the Electrical Conduction Produced By It," Philos. Mag. 47, 109 (1899).
[2] E. Rutherford, "The Structure of the Atom," Philos. Mag. 27, 488 (1914).
[3] J. J. Thomson, "On the Structure of the Atom: an Investigation of the Stability and Periods of Oscillation of a Number of Corpuscles Arranged at Equal Intervals Around the Circumference of a Circle; with Application of the Results to the Theory of Atomic Structure," Philos. Mag. 7, 237 (1904).
[4] E. Rutherford, "The Scattering of α and β Particles by Matter and the Structure of the Atom," Philos. Mag. 21, 669 (1911).
[5] H. Geiger, "On the Scattering of the α Particles by Matter," Proc. R. Soc. A 81, 174 (1908).
[6] H. Geiger and E. Marsden, "On a Diffuse Reflection of the α-Particles," Proc. R. Soc. A 82, 495 (1909).
[7] H. Geiger, "The Scattering of the α Particles by Matter," Proc. R. Soc. A 83, 492 (1910).
[8] E. Rutherford, "The Origin of α and β Rays From Radioactive Substances," Philos. Mag. 24, 453 (1912).
[9] H. Geiger and E. Marsden, "The Laws of Deflexion of α Particles Through Large Angles," Philos. Mag. 25, 604 (1913).