 |
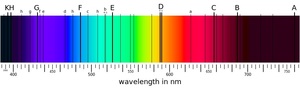
| Fig. 1: Illustration of the solar spectrum. The thin black bands are absorption lines corresponding to the elements contained in the sun's outer atmosphere. (Source: Wikimedia Commons) |
Nucleosynthesis is the process by which atoms of lighter chemical elements fuse together, creating atoms of heavier elements. [1-3] Atoms are comprised of three elementary particles - protons and neutrons bound into a dense nucleus and electrons surrounding that nucleus. In the fusion process, light nuclei collide, recombine their protons and neutrons into heavier nuclei, and release energy. This process requires tremendous amounts of heat and energy; as such this fusion can only happen in extreme environments. [4]
This fusion of nuclei in stars is the most concrete theory today for the origin of heavy elements. Our current understanding is that the early universe contained only very light elements, chiefly the single-proton hydrogen atom. The remaining elements in the periodic table, including those essential for the molecules in our bodies, were then created in stars through nucleosynthesis before the earth was formed.
One of the strongest pieces of evidence for the theory of stellar nucleosynthesis is the observation of absorption lines of the element Technetium from distant stars.
The first discoveries in the field of optics were fundamental to the science of spectroscopy that enabled the measurements of Technetium identification. Isaac Newton used a prism to demonstrate that white light could be split into components of colored light, or light of different wavelengths. [5] Joseph von Fraunhofer later achieved the same feat using a diffraction grating, another optical tool that more precisely splits light into its component "colors." This allowed for exact numerical measurement of the optical wavelengths of light. When Fraunhofer used a diffraction grating to observe the light coming from our sun, he discovered the first "stellar spectrum," an illustration of which is shown in Fig. 1. This consists of a set of "dark lines" missing from Newton's rainbow. [6]
Building on these initial discoveries, correlation between the wavelengths produced and element composition of a light source was discovered. Gustav Kirchhoff and Robert Bunsen used Fraunhofer's diffraction grating to analyze the unique wavelengths of light emitted by various elements. Their experiment involved "exciting" molecules and elements by heating them over a flame. Once energized, element would emit the heat energy it absorbed from the flame at specific wavelengths of light unique to the element - their chemical signatures. Recording the emission lines using a diffraction grating, they created a template for the wavelength "fingerprints" for the comprehensive list of elements they tested. [7]
This experimental groundwork outlining correlation between specific elements and specific wavelengths laid the foundation for Kirchhoff and Bunsen's discovery that the wavelength bands missing from the solar spectrum - the absorption lines Fraunhofer found - correlated with emission lines of elements they had tested in the lab. The spectral lines of the sun (Fig. 1) directly represented the elements that comprised it. [7,8]
As all stars emit these specific wavelength "fingerprints", astronomers to this day use the absorption lines of stars and planets to identify the specific atoms and molecules that comprise them. The absorption lines amount to a wavelength "fingerprint" for an individual element. This method, called spectroscopy, was fundamental to the first discovery that supported nucleosynthesis.
 |
| Fig. 2: Photograph of the Carina Nebula. Note the stars with a reddish hue. Some of these are red giants, stars similar to the ones where Technetium's absorption lines were first found. (Courtesy of the European Southern Observatory. Source: Wikimedia Commons) |
The nature the element Technetium's decay also offers important evidence for the rationale behind nucleosynthesis. Technetium is the lightest element that is exclusively radioactive, having no stable isotopes. As it is unstable, its nucleus decays spontaneously to the nucleus of lighter elements, turning a Technetium atom into a different atom of a more stable element. [9]
The half-life of a radioactive element, like Technetium, is a measure of the amount of time that radioactive decay takes - specifically how long it takes for half of a sample to radioactively decay.
Scientists empirically determined the half life of Technetium by carefully analyzing the decay of a portion of Technetium containing molecules in a lab. This process first involved the collection and isolation of materials containing the element en masse. Once a sample of 100% Technetium-containing molecules was purified from nuclear waste sources, the sample was left to decay for a carefully measured amount of time. As Technetium decayed to other elements in this period, the compounds containing the resultant stable atoms can be dissolved and removed with certain chemical solvents. Dissolving and removing the decayed portions of Technetium created a measurable enough change in mass to determine the fraction of atoms that had decayed. By analyzing the proportion of material decayed per amount of time, scientists were able to determine the amount of time needed for half the Technetium to decay. As a result they deduced the longest possible half life for a Technetium atom to be 4.2 million years. [10,11]
In 1952, astronomer Paul W. Merrill used the process of spectroscopy to find four spectral lines of Technetium atoms in red giant stars (Fig. 2) estimated to be billions of years old. [12] As Technetium's absorption lines were clearly present in several star's spectrum, it followed that the stars contained the radioactive element that - based on its half life - could not have been present in the stars composition when the star was born.
The discovery was strong evidence that Technetium had to have been synthesized recently within the star. [13,14] In 1957, a landmark paper by Burbidge et al. titled "The Synthesis of the Elements in Stars" used these observations to outline the process of stellar nucleosynthesis as understood today. [15] This also set the groundwork for understanding other instances of nucleosynthesis during the Big Bang and supernovae that created much lighter and much heavier elements on the periodic table.
With the direct observation of Technetium's spectral lines in stars, an understanding of spectroscopy, and the half life of the longest living isotopes of the element, this process that powers stars and creates the elements falls into place. Stellar nucleosynthesis is the answer to the common cosmic origin of the building blocks that comprise you and me.
The atoms that comprise you, me, and the universe around us were forged in stars.
© George Sivulka. The author grants permission to copy, distribute and display this work in unaltered form, with attribution to the author, for noncommercial purposes only. All other rights, including commercial rights, are reserved to the author.
[1] H. Geiger, "On the Scattering of the α-Particles by Matter," Proc. R. Soc. A 81, 174 (1908).
[2] J. Chadwick, J. E. R. Constable, and E. C. Pollard, "Artificial Disintegration by α-Particles," Proc. R. Soc. A 130, 463 (1931).
[3] J. J. Thomson, "Cathode rays," Philos. Mag. 44, 293 (1897).
[4] H. A. Bethe, "Energy Production in Stars," Phys. Rev. 55, 434 (1939).
[5] I. Newton, Opticks: Or a Treatise of the Reflections, Refractions, Inflections, and Colours of Light (Dover Publications, 2012).
[6] J. Fraunhofer, "Bestimmung des Brechungs- und Farben-Zerstreuungs-Vermögens verschiedener Glasarten, in Bezug auf die Vervollkommnung achromatischer Fernröhre," Denkschr. Ak. Münch. 5, 193 (1817).
[7] G. Kirchhoff, "Über die Fraunhoferschen Linien," Ann. Phys. 185, 148 (1860).
[8] G. Kirchhoff, Researches On the Solar Spectrum, and the Spectra of the Chemical Elements (Nabu Press, 2012).
[9] C. Perrier and E. Segré, "Technetium: The Element of Atomic Number 43," Nature 159, 24 (1947).
[10] G. Audi et al. "The NUBASE Evaluation of Nuclear and Decay Properties," Nucl. Phys. A 729, 3 (2003).
[11] P. W. Merrill, "Technetium in the Stars," Science 115, 484 (1952).
[12] C. Hamman, "Technetium of the Stars," Physics 241, Stanford University, Winter 2011.
[13] G. Wallerstein et al. "Synthesis of the Elements in Stars: Forty Years of Progress," Rev. Mod. Phys. 69, 995 (1997).
[14] E. M. Burbidge et al. "Synthesis of the Elements in Stars," Rev. Mod. Phys. 29, 547 (1957).