 |
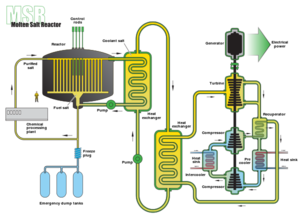
| Fig. 1: Diagram of a molten salt reactor. (Source: Wikimedia Commons) |
Recently emerging from a long drought in attention by various governments and scientific interests, Thorium nuclear fuel cycles show a flicker of promise for those interested in the future of nuclear energy. Thorium has lingered in the periphery of nuclear energy since World War II, after which its development was put on halt in favor of U-235 processes. Th-232, while not a fissile substance outright, can be transmuted to fissile U-233, which can be used as a fuel. Additionally, a newer type of reactor known as a Liquid Fluoride Thorium Reactor, which chiefly uses Thorium and U-233 in a Fluoride based salt mixture, continues to garner attention as a cleaner, safer alternative to the more common U-235 based reactors (see Fig. 1).
Perhaps Thorium's most significant advantage over uranium is its relative abundance. Thorium has been estimated to be approximately three times more abundant in the Earth's crust than uranium. Thorium is more common than tin, mercury, and silver. [1] With few industrial and common applications, Thorium is effectively one of the world's most energy-dense unexploited resources. Though thorium requires a small amount of fissile material to begin its process of energy production, the exploitation of Thorium-based fuel processes could extend the useful life and preservation of the earth's nuclear fuels for generations to come.
As with any nuclear fuel, Thorium possesses a potential for misuse in the production and proliferation of nuclear weapons. However, compared to the commonplace U-235 fuel cycle, Thorium augmented fuel cycles can be argued to be preferable from a non-proliferation standpoint. A conventional U-235 light-water reactor produces Pu-239, a chemically distinct and weaponizable isotope, by irradiating U-238. The remaining Pu-239 is easily separable from U-238 by simple chemical means, and thus presents a proliferation risk. The thorium fuel cycle presents an opportunity to mitigate this risk with introduction of non-fissile, chemically similar isotopes.
As mentioned earlier, the Rhorium fuel cycle produces fissile U-233, which presents a proliferation risk. However, if the initial thorium fuel source is intermixed with "garbage" U-238, the resulting U-233 will not be separable by the simple chemical means by which fissile Pu-239 can be separated from depleted U-238. The U-233 will be intermixed with U-238, making the harvesting of fissile U-233 significantly more difficult. Additional substances can be added to the fuel in order to make leftover Pu-239 more difficult to separate out, further inhibiting the ambitions of would-be proliferators.
Thorium is not without its share of storage and environmental concerns once the fuel has completed its useful life. Current uranium-thorium hybrid processes are hampered by the disposal of U-233, which always contains traces of U-232 and its gamma-emitting daughter nuclides. Commonplace U-235 processes are hampered by this effect as well with traces of Pu-239, though for both fuel cycles this has the advantage of adding to proliferation resistance by making the spent fuel harder to handle and easier to detect. Additionally, Thorium fuel cycles and commonplace Uranium-Plutonium cycles all produce radioactive fission products, the primary to cheap and easy disposal, in approximately the same amounts. [2]
Thorium shows some amount of promise as an abundant and potentially eventually cost effective augmentation to traditional uranium-based fuel cycles. Thorium has numerous useful qualities, most saliently its abundance, which give it a fighting chance for its place as a fuel of the nuclear future. However, the timing of thorium's transition into this role remains unclear.
© Erik Holmvik. The author grants permission to copy, distribute and display this work in unaltered form, with attribution to the author, for noncommercial purposes only. All other rights, including commercial rights, are reserved to the author.
[1] R. Hargraves and R. Moir, "Liquid Fluoride Thorium Reactors: An Old Idea in Nuclear Power Gets Reexamined," Am. Sci. 98, 309 (2010).
[2] W. D. Loveland, D. J. Morrissey, and G. T. Seaborg, Modern Nuclear Chemistry (Wiley, 2006), p. 317.