 |
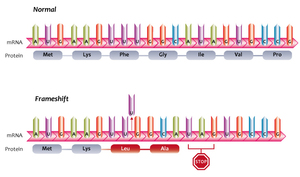
| Fig. 1: A frameshift mutation in DNA that results in an early termination of codon translation into protein. (Source: Wikimedia Commons) |
In an effort to better understand the intricate, physical nature of random, disease-inducing mutations, the field of quantum biology - which harmoniously bridges the principles of quantum physics to biological systems - aims to study evolutional phenomena in the classical world. Just a century ago, the notion of combining the mathematical proofs of quantum mechanics with the ever-differentiating phenotypes in biology would have seemed like a mismatch, at best. The dawn of interdisciplinary research has changed such notions. Advances made by Erwin Schrdinger in his book "What is Life?", as well as the later discovery of the DNA helix model by Watson and Crick, paved the way for physicists and biologists to openly collaborate.
To observe the potential quantum phenomena that take place in the very base pairs of our DNA, biologists must first get on the right scale. Biology has largely focused on non-nanoscale studies of bacteria, viruses, and DNA (10-6 m, 10-7 m, 10-8 m, respectively); however, quantum alterations start at a much smaller scale. How small exactly? Think 10-15 (or one femtometer), the size of the very nucleus at the center of an atom.
The critical question remains, however, as to what extent these changes in the very subunits of our genetic code are attributed to quantum effects versus biological phenomenon.
While quantum principles generally apply to particles of atomic or subatomic size, biological systems are made up of trillions of atoms interacting with each other in a host of unique, complex, and disparate ways. [1] So how can these two vast disciplines be linked together? With respect to disease and random mutations, the first thought is to look at the very information that encodes for humans' traits: DNA. By first understanding the mechanism of how mutation occurs, scientists believe that they can crack the code on why our DNA may be susceptible to such random mutations and, subsequently, genetic disease.
Human DNA is made out of four base pair nucleotides: Adenosine (A), Tyrosine (T), Guanine (G), and Cytosine (C), where Adenosine complementary pairs with Tyrosine and Guanine with Cytosine. The DNA double helix is held together by hydrogen bonds serving as the proverbial glue between each base pair. As these base pairs are transcribed from DNA into RNA, and as the RNA is translated into protein, these base pairs (with DNA's T base pair swapped for RNA's Uracil (U) base pair) are read in groupings of three, which are referred to as codons. These codons provide the code for start and stop sites as well as a host of proteins that execute different tasks in our body. When mutations occur in the coding region of the DNA - that is, the portion of DNA that will eventually encode for protein after transcription - the alteration could lead to devastating consequences.
An important statistic that geneticists keep note of is the likelihood of such mutations occurring in the genome. It is estimated that for every 10,000 to 100,000 bases copied by DNA polymerase, there is a mistake made that, if left uncorrected, may lead to devastating consequences. [5] This is particularly true when the mutation occurs within the coding region of DNA, which will be transcribed into RNA and turned into protein. A non-silent point mutation of a base in this region is the culprit of diseases like cystic fibrosis or Tay-Sachs disease, among many others. Such mutations can also give rise to more common diseases among the population. For example, one small mutation in an autosomal cell can lead to the development and proliferation of cancer (see Fig. 2).
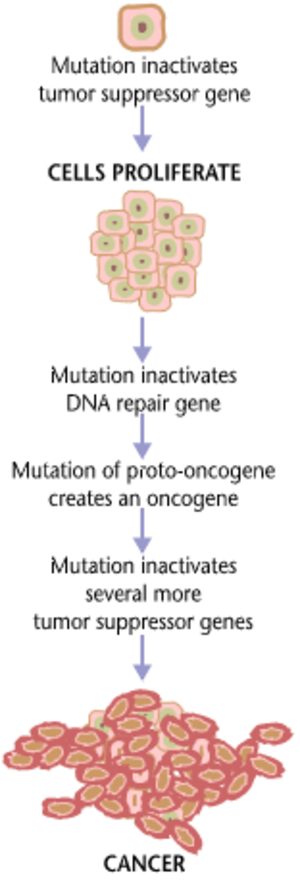 |
| Fig. 2 The progression of cancer: from DNA mutation to cell proliferation. (Source: Wikimedia Commons) |
With respect to diseases like cancer, there are several types of mutations that are of particular interest to quantum biologists: insertions and deletions. Insertion or deletion of a base pair from the coding region often results in a frameshift mutation, in which the reading frame is shifted and the original codons are all changed by one (or more) nucleotide(s). An example of a DNA frameshift mutation is shown in Fig. 1. If lucky, the frameshift may be a silent mutation, in which the codons code for the same original protein or are outside of the start- stop coding region. For all too many patients, however, detrimental frameshift mutations can give rise to life-threatening diseases and conditions.
Such random mutations fascinated theoretical physics Professor Jim Al-Khalili and Molecular Genetics Professor Johnjoe McFadden at the University of Surrey. These scientists posit a quantum theory behind how mutations in our DNA arise: that a single nucleotide, as a single unit, is subject to quantum strangeness. Professor Al-Khalili mentions in a Discover Magazine interview mentions that their notion was "hand-waving, but we had an inkling that something quantum was happening at the level of DNA". [2] After observing an E. coli model with respect to lactose tolerance/intolerance, the pair contended that, before binding, the nucleotide in question exists in both a mutated and normal state on the DNA helix; and from this theory, a parallel can be made to the Principle of Superposition, which states that all possible states that the particle may be in exist simultaneously. Further, the principle suggests the idea that particles may exist in two places at once, an oddity of quantum physics. [3]
These experiments led to a host of other institutions behind the intricate cellular processes that allows organisms to function, including plant photosynthesis, carbon transfer, and even European robin migration. [4] Importantly, these very quantum explanations for mutation grant scientists insight as to how mutation and disease may occur.
As is the case with every budding field in science, there is a great deal of scrutiny that must be applied to initial studies after the excitement settles. Taking these "hand-waving" proposals into perspective will allows us to determine the validity and potential of such research. There are certain biological phenomena - such as photosynthesis or bird navigation patterns - that would certainly benefit scientists understanding if parallels could be drawn between these biological mechanisms and quantum theories. [4] However, not only are these propositions far from conclusive, but they "may simply show that reality always appears quantum in nature, if you look closely enough". [6] A comprehensive overview of research indicates that many publications in the field of quantum biology lack numerical support for their hypotheses or fail to produce undisputed evidence of the role of quantum theory in microevolution of cells or erroneous DNA replication.
If nature has indeed not discovered a method to exploit quantum mechanics, the critical follow-up question is: why not? With respect to DNA mutations, the classical understanding is that mutations arise via a hereditary (in which mutations are passed down the germline) or somatic (acquired mutations that cannot be passed down) pathway. [7] Therefore, causes of mutation have historically centered at the molecular level, where cellular machinery that creates the DNA sequence makes a copy, transcription, or translation error that results in a genetic polymorphism or mutation. The cell has developed many methods of fixing such errors in the genetic code of life; in fact, 99% of mutations and mismatched nucleotides are "proofreading" mistakes. [8] These very errors are caused by the addition of an incorrect nucleotide to a strand of DNA, which ultimately stalls replication since the 3' (3 prime)-OH end of the DNA is in an incorrect position. DNA polymerase - an enzyme that creates DNA out of these nucleotides - does a remarkable job at ameliorating most, but not all, of these proofreading errors in a process called mismatch repair. [8] At the heart of these mutations lies the intrinsic fact that the DNA helix is flexible, and therefore can allow these errors to arrive. [7] The argument can thereby be made that mutations are derived from innate, flawed structural characteristics of the double helix rather than quantum shifts in the base pairs themselves.
If the DNA polymerase does in fact miss the error in the nucleotide sequence - as it does for approximately one out of every 100,000 nucleotides - and the flawed base is within the RNA's protein coding sequence, such errors can result in concerning mutations. [8] At this point, the mutation persists throughout the DNA, RNA, and potentially protein sequences because of micro-level inaccuracies in the cell and an imperfect enzyme error correction mechanism, rather than a quantum level phenomenon. These mutations have the potential to cause diseases like cancer, where the replication and propagation of such flawed sequences can lead to its proliferation throughout the body, as seen in Fig. 2.
Overall, the very intercellular mechanisms that lead to the rise of such harmful mutations are thoroughly reported and confirmed. The strong evidence detailing helix flexibility or mismatch repair casts a nebulous shadow upon quantum biology. Perhaps what quantum biology researchers have truly found is that mutations in our genetic code reflect quantum theories, rather than having innately quantum processes.
The field of biology has only seen the potential for quantum physics as applied to low-level cell machinery and genetics. Scientists have yet to benefit from exploiting these very quantum tools as proposed by quantum biology researchers, largely due to the nature of this research being highly theoretical. As further collaborations and funding are invested in the interdisciplinary studies of quantum biology, physicists and biologists must ensure that their findings are viewed with a critical eye and in context of previous, researched-backed theories of micro-level cellular machinery being responsible for DNA mutation, evolution, and repair. Finally, the added use of computational and mathematical models to simulate how such quantum changes in DNA can lead to in-vivo mutations are fundamental to cementing the future of quantum biology as a fundable research venture.
© Persiana Saffari. The author warrants that the work is the author's own and that Stanford University provided no input other than typesetting and referencing guidelines. The author grants permission to copy, distribute and display this work in unaltered form, with attribution to the author, for noncommercial purposes only. All other rights, including commercial rights, are reserved to the author.
[1] J. Al-Khalili and J. McFadden, "You're Powered By Quantum Mechanics. No, Really," The Guardian, 25 Oct 14.
[2] Z. Merali, "Solving Biology's Mysteries Using Quantum Mechanics," Discover Magazine, 29 Dec 14.
[3] A. S. Sanz and S. Miret-Artes, A Trajectory Description of Quantum Processes. II. Applications, (Springer, 2014).
[4] J. Palmer and A. Mansfield, "Quantum Biology: Do Weird Physics Effects Abound in Nature?," BBC News, 28 Jan 13.
[5] I. J. Kimsey et al., "Visualizing Transient Watson-Crick-Like Mispairs in DNA and RNA Duplexes," Nature 519, 315 (2015).
[6] "The Quantum Biology Conundrum," Technology Review, 8 May 12.
[7] L. H. Hartwell et al., Genetics: From Genes to Genomes, 5th Ed. (McGraw-Hill Education, 2014),
[8] L.A. Pray, "DNA Replication and Causes of Mutation," Nature Education 1, 214 (2008).