 |
| Fig. 1: Lithium Battery Charging (Source: Wikimedia Commons) |
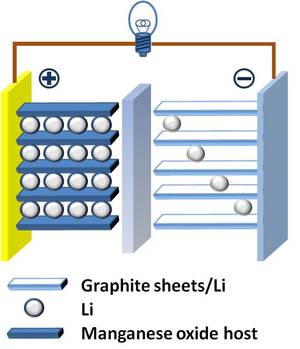
The lithium battery was invented in 1912, by physical chemist Gilbert Lewis. His experimentation with lithium-based batteries would eventually replace nickel-cadmium batteries, which had been the go to battery for portable equipment. Lithium is a good candidate for a portable battery for a couple of reasons: it is the lightest of all chemical metals, and it has a high energy density, resulting in lithium having a high electrochemical potential. [1] Despite Lewis experimenting with a lithium-based battery in 1912, lithium batteries were not commercialized for public use until 1970. This is due to the fact that the lithium metal is naturally unstable, which became problematic when recharging an energy storage device using lithium batteries. [2] As a result, batteries consisting of lithium alone can not be recharged. In 1970, the lithium-ion based battery was commercialized. The lithium-ion version of the portable battery proves to be more stable than lithium alone, with the trade-off of a loss in total energy capacity, but the lithium-ion battery can be recharged. There are now a number of different types of lithium-based batteries, in attempts to improve energy density and reduce the risk of complications with the battery. The energy density of a lithium-ion battery is typically [3]
Generally, lithium batteries are typically reliable and productive with no issues. However, when issues with lithium-based batteries do occur, the failure is usually due to a short circuit within the charging cell of the battery. [2] When a short circuit does occur, the majority of the time the short circuit is caused by microscopic metal particles coming into contact with the other parts of the battery cell. Battery cells that separators of 24 micrometers or smaller are at risk of outside particles contaminating the cell. Battery manufacturers design their batteries to limit short circuits as described above by focusing on the design of electrolyte, separator and electrode. [2] Additionally, lithium batteries can overheat. The process of overheating begins with a number of microscopic particles in one distinct spot. There is an electric current between the location of the particles and the electrodes of the battery cell, heating the location to 500°C, and greatly increasing the risk of an explosion. Exercise caution when dealing with an overheated lithium battery and keep it away from any flammable or combustible materials.
In addition to correcting engineering flaws in reducing incidents with lithium battery explosions, management oversights are also the source of some of these incidents. There are a number of commercialized products which use defective lithium batteries that work fine at first, but over time, the defective battery eventually fails. These products can be released by manufacturers, despite not being ready for public use. There is a documented history of issues with lithium batteries, such as Samsung recalling a large number of their phones due to defective batteries.
© Sam Pfeil. The author warrants that the work is the author's own and that Stanford University provided no input other than typesetting and referencing guidelines. The author grants permission to copy, distribute and display this work in unaltered form, with attribution to the author, for noncommercial purposes only. All other rights, including commercial rights, are reserved to the author.
[1] B. Scrosati and J. Garche, "Lithium Batteries: Status, Prospects and Future," J. Power Sources 195, 2419 (2010).
[2] K. Weikert, "Lithium Battery Dangers: Past and Present ," Physics 240, Stanford University, Fall 2016.
[3] M. Armand and J. M. Tarascon, "Building Better Batteries," Nature 451, 652 (2008).