
 |
| Fig. 1: A plutonium fuel pellet (Source: Wikimedia Commons) |
Nuclear batteries have been used successfully in space exploration for over four decades, powering twenty-seven missions. The newest model, the Multi-Mission Radioisotope Thermoelectric Generator (MMRTG), will power the future Mars 2020 Rover. [1] These nuclear batteries have been chosen for space applications time and again because, despite their cost and the waste they generate, they are much longer lasting and can survive in much harsher conditions than their chemical counterparts. Both are key features for space missions expected to last multiple decades in the vacuum of space. The hardiness of the RTG battery model is evidenced by the fact that in twenty-seven missions, no RTG has failed. The Voyager II even continued to transmit data forty years after its launch date, demonstrating a lifespan no chemical battery or photovoltaic cell could hope to match. [2] However, until recently, nuclear batteries have rarely seen applications outside of space exploration. Besides their use in pacemakers in the 1970s, they have been avoided due to their high risk and cost. This changed in 2009, when a new breakthrough in nuclear battery technology at University of Missouri sparked the interest of the U.S. military. [3] They began pouring funds into the project. If their investment pans out, it could pave the way for the use of nuclear batteries in an even wider variety of fields.
Radioactive thermoelectric generators convert heat energy into electricity. Specifically, they convert the heat released by the nuclear decay of radioactive isotopes. The typical isotope used for RTGs is Plutonium-238, in the form of a plutonium dioxide fuel pellet which can be seen in Fig. 1. Strontium-90 is also fairly common. The choice of isotope depends largely on its possession of three characteristics: a long-half life; low gamma radiation; and high power and energy densities. Stability and a high melting point also factor into the decision. Both of these increase the inherent safety of the RTG.
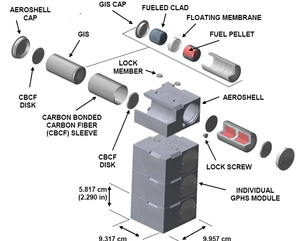 |
| Fig. 2: A diagram of an individual GPHS module (Source: Wikimedia Commons) |
Safety is addressed in the fuel encapsulation design, called the General Purpose Heat Source (GPHS) module, which can be seen in Fig. 2. The GPHS module blocks the beta and gamma radiation of the fuel pellets using a series of layers: a fuel cladding; a graphite impact shell; a carbon-bonded carbon fiber insulation; and a fine weave pierced fabric aeroshell. [4] Since the plutonium pellets release primarily alpha particles, they require only 2.5 mm of lead shielding, making the precautions built into the GPHS module more than enough. The MMRTG designed for the Mars 2020 rover will use eight individual GPHS modules stacked on top of each other, as compared to the previous module which used eighteen. Recent improvements in material allowed for this downsizing, while still maintaining a power output of 110 watts. [1]
The RTG design uses a static thermoelectric energy converter to convert heat to electricity. These converters produce electricity from heat using the Seebeck effect, whereby thermoelectric materials experiencing a gradient in temperature produce a voltage. This voltage then sends current through the material, in a direction dependent on how the material is doped. When the thermoelectric material is connected in series with a load, current passes through the load and produces useful power. While the process is both reliable and compact, and generates no vibration, noise, or torque, it is much less efficient than dynamic conversion processes. Most thermoelectric energy converters operate at less than ten percent efficiency. [4] However, this seemingly low efficiency still compares favorably with static chemical batteries, since RTGs have much longer lifespans and higher energy densities. Some nuclear batteries have energy densities as high as 4,500 watt-hours per kilogram, as compared to the typical batteries powering today's electronics, lithium ion batteries, which produce between 110 and 160. [5]
Nuclear batteries are ideal for any application in which the device cannot be routinely maintained and serviced, or must withstand extreme environmental conditions. Many devices fall under this category, including those used in deep sea exploration, in implanted medical devices, and in front-lines military operations. An example of the latter is that soldiers must carry nearly thirty pounds of chemical batteries in order to keep their radios and sensors maintained. During the summer, the added weight can hasten the onset of potentially deadly heat exhaustion. Nuclear batteries have the potential to eliminate the burden of up-keeping such devices and therefore save lives. Chemical batteries also make it difficult to power the electric circuits responsible for protecting missiles and planes from tampering, since those circuits are expected to last for long periods and withstand temperatures ranging from -65°C to 150°C. In addition, the circuits experience high-frequency vibrations, high humidity, and blasts of salt. [3] Nuclear batteries are a very viable option in this case because the memory chips that control such anti-tamper systems require relatively low continuous power over a long period of time, reducing the drawback of having an inefficient battery.
Seeing the potential for batteries that could address these situations, an associate professor at University of Missouri, Jae Kwon, developed a new design, called a betavoltaic cell. Each cell is a tiny chip constructed by mixing the chosen radioactive isotope, in powder form, with a selenium based semi-conductor material, and then melting the two together. This increases the efficiency by allowing radiation from all directions to strike the semiconductor. It also eliminates any danger of the beta radiation destroying the semiconductor, since the material remains in a liquid state and thus has no crystalline structure to damage. [6] Furthermore, the design incorporates several layers of aluminum to ensure the protection of anyone interacting with the cell. [3] The betavoltaic cell caught the attention of Lockheed Martin, which began testing the betavoltaic cell for U.S. military use beginning in 2009. [3] The cell still has a long way to go before it produces enough power to run most devices, but hopefully military funding will help pave the way to a solution. Military funding could also bolster efforts to find cheaper heat sources, which would cut down the cost and attract companies from other fields. Thus the success of this endeavor could allow for the realization of a potential in nuclear batteries that has so far remained largely unexplored.
© Isabel Gueble. The author grants permission to copy, distribute and display this work in unaltered form, with attribution to the author, for noncommercial purposes only. All other rights, including commercial rights, are reserved to the author.
[1] R. Bechtel, "Multi-Mission Radioisotope Thermoelectric Generator," U.S. National Aeronautics and Space Administration, October 2013.
[2] J. Margolis, "40 Years and Counting: The Team Behind Voyager's Space Odyssey," The Guardian, 15 Mar 15.
[3] K. Bourzac, "A 25-Year Battery," MIT Technology Review, 17 Nov 09.
[4] G. R. Schmidt, T. J. Sutliff, and L. A. Dudzinksi, "Radioisotope Power: A Key Technology for Deep Space Exploration," in Radioisotopes - Applications in Physical Science, ed. by N. Singh (InTech, 2011), p. 419.
[5] N. Edmonson, Technological Foundations of Cyclical Economic Growth, ed. by N. Edmonson (Transaction Publishers, 2009), p. 264.
[6] B. H. Kim and J. W. Kwon, "Plasmon-Assisted Radiolytic Energy Conversion in Aqueous Solutions," Scientific Reports 4, 5249 (2014).