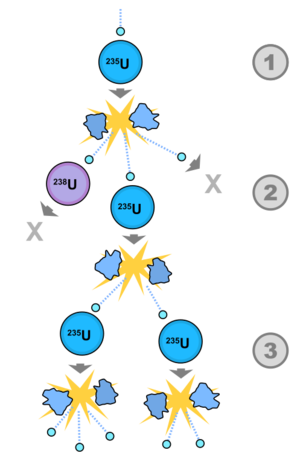
 |
| Fig. 1: Chain reaction by neutrons inducing fission. (Source: Wikimedia Commons). |
Nuclear power is not renewable - the amount of uranium on the planet is finite. Based on current burn rates with the standard reactor design, the lifetime is surprisingly short. We can take the optimistic estimate of the worldwide reserves at 30 x 106 metric tons of natural uranium at a cost of $1000 per kilogram. [1] Approximating 500 reactors worldwide that require about 200 metric tons of natural uranium per year to operate at current efficiencies, the reserves would last for 300 years. If nuclear power is to supply the entire world, this decreases the lifetime by a factor of 20. If we want nuclear power to continue to provide electricity, it only makes sense to use this supply efficiently. The most common "light water" nuclear reactor design only burns up about 5% of the available fuel before labelling it waste and discarding it. This is done because the buildup of fission product waste (such as Kr and Xe) eats the neutron population, and the fissile uranium fuel is mostly burned. Additionally the light water reactor requires uranium enrichment where only 1/10 of the ore from the ground makes it into the fuel and trailings of depleted U-238 are not used. U-238 and Th-232 atoms are about 150 and 450 times more abundant than U-235 and can also be used as fuel. [2] By developing advanced reactors and waste processing methods to efficiently burn uranium and thorium, we can therefore extend the lifetime of nuclear power by over a factor of 100.
Breeder reactors are capable of burning a larger fraction of the available energy by creating new fissile material from the fertile material during operation. A serious drawback of breeders is safety; using fast (high energy) neutrons to efficiently breed means water cannot be used to slow down neutrons. This removes a crucial safety mechanism where the evaporation of water from the core shuts off the chain reaction. Heavy water reactors offer a similar failsafe to light water reactors but with a better neutron economy due to the low absorption by deuterium. The question we would like to ask in this report is whether or not a heavy water reactor is capable of breeding new fuel successfully to combine benefits of the thermal reactor's failsafe and breeder reactor's fuel efficiency. To answer this question, we will review the moderation of the neutron population, the design of thermal reactors, and the process of breeding fissile fuel.
A nuclear reactor is said to be critical when the neutron population remains constant and the reaction is self-sustaining. nucleus absorbs a neutron and fissions. The nucleus separates into two fission products and sheds prompt, fast neutrons. The neutron-rich fission products can either decay by releasing a beta (electron) or delayed neutrons to continue the process. The fission cross section for fissile nuclei diverges as σf ~ 1/E1/2 at low energies, so "thermal" reactors are designed with a moderator that slows down the neutron population while absorbing as few as possible. By enriching the fuel (increasing the amount of fissile isotope), the neutron released from a fission event is more likely to hit another fissile nucleus and thereby create a chain reaction. By choosing the moderating material wisely, the enrichment requirement can be brought down to natural uranium ore at 0.72% enrichment for heavy water reactors. Since fertile U-238 can absorb a neutron and transmute into fissile Pu-239 after two beta decays, the absorbed neutron can also go towards generating new fuel. The absorption cross section for fertile nuclei contains sharp peaks in the "epithermal" region between eV and MeV energies due to resonances between the incident (plus binding) energy of the neutron and the excited states of the nucleus (plus neutron). By designing the reactor to maximize the neutron flux over this region, the neutron has the opportunity to hit a resonance and effectively breed new fuel.
|
|||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Table 1: Neutron scattering and absorption cross sections in units of barns = 100 fm2 = 10-28 m2 at E = 0.0253 eV = 293 K, average logarithmic energy loss ξ, number of collisions to thermalize N, and moderating ratio MR. [3] |
We can consider a simple elastic scattering problem of a incident neutron of mass m=1 and energy E on a stationary nucleus of mass M=A where A is the atomic number. The ratio of the neutron energy before and after is E'/E = (v')2/(v)2 = [(1+α)+(1-α)cos(θ)]/2 where α=(A-1)2/(A+1)2 and cos(θ) is the angle the neutron was scattered into in the center of mass frame. [4] The average logarithmic energy loss ξ is given by:
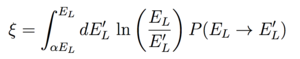 |
(1) |
where (for isotropic scattering) the probability a neutron scatters from energy EL to an energy interval of width dEL' around EL' is given by P(EL→EL') dEL' = [(1-α)EL]-1 dEL' and the integral is taken from the maximum energy transferred αEL when θ = π. [4] Plugging this in, we obtain
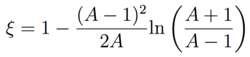 |
(2) |
The number of scatterings N to thermalize the neutron is determined by E'=eN ξ E where E' ~ 1 eV ~ 11,600 K is the "thermal" energy scale and E ~ 2 MeV is the fast neutron birth energy scale. Some values for ξ and N are show in Table 1 for different elements. The moderating ratio is a measure of the effectiveness of a moderator in reducing the number of collisions to thermalize while not absorbing the neutron population. The moderating ratio is defined as MR = ξ (σs/σa) where σa is the neutron absorption cross section and σs is the neutron scattering cross section. For materials with multiple atomic species we take the average:
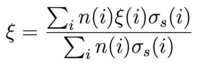 |
(3) |
where σs(i) and n(i) are the scattering cross section and number density respectively for species i. [4] The moderating ratio is then MR = ξ (Σs/Σa) where Σa = Σi n(i)σa(i) is the macroscopic neutron absorption cross section and Σs = Σi n(i)σs(i) is the macroscopic neutron scattering cross section. Some values of the moderating ratio are shown in Table 1 for different elements. The density of light water is nH2O = 3.35 × 1022 cm-3. We have nH = 2 × nH2O and nO = 1 × nH2O. Similarly for heavy water, nD2O = 3.68 × 1022 cm-3, nD = 2 × nD2O and nO = 1 × nD2O. Using the values from Table 1, the moderating ratios (density cancels) are then given by MRH2O = 249 and MRD2O = 10,167. We therefore see that while hydrogen has a larger scattering cross section, the lower absorption cross section of deuterium results in a higher moderating ratio.
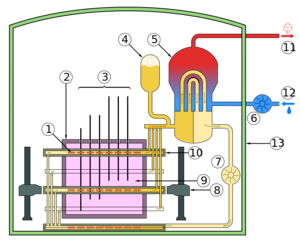 |
| Fig. 2: Schematic for heavy water CANDU reactor. (Source: Wikimedia Commons). |
Light water reactors use H2O as both the neutron moderator and coolant to carry heat away from the core. This primary loop is connected to an intermediate loop or directly to a steam cycle loop with turbine to produce electricity. High pressures on the order of 100 atmospheres require additional safety considerations since a leak could spread radioactive material that worked its way into the water. Light water reactors require replacement of fuel rods about every 4 years due to the build up of neutron-eating fission products such as samarium and xenon. Xe-135, produced from the decay of I-135, has a thermal absorption cross section (in units of barns = 100 fm2 = 10-28 m2) of σa(Xe-135) = 2.6 x 106 barns compared with the U-235 fission cross section of σf(U-235) = 580 barns at E = 0.0253 eV = 293 K. [3,4] The uranium enrichment requirement for light water reactor fuel is about 3-5%. While most of the initial fissile fuel has been used up by the time of replacement, over 90% of the energy content remains in the form of fertile fuel. Safety mechanisms including control rods made of neutron absorbing materials such as boron or cadmium which are suspended by electromagnets. If the power goes out or a switch is tripped, the rods fall into the core under gravity and quickly eat the neutron population to bring the core below criticality. If the control rods aren't effective, there is a built-in failsafe in the water itself: if the water moderator boils away, the neutrons won't be thermalized effectively and the cross section for inducing fission remains low.
Heavy water reactors, such as the Canadian Deuterium Uranium (CANDU), are also a thermal reactor, but use heavy water D2O as a moderator. The heavy water in the CANDU reactor is contained in an unpressurized, 7 meter diameter tank setup called the "calandria" which contains 380 of horizontal high pressure zirconium alloy "calandria tubes" consisting of 12 uranium oxide fuel pellets and D2 coolant as shown in Fig. 2. [4,5] The fuel bundles in the core contain 100,000 kg of natural uranium and heat coolant from 265°C to 310°C which exchanges heat with a light water steam power cycle. While fuel bundles need to be changed every 1-2 years, the design allows for changing individual bundles by pushing new fuel into the tube and removing old fuel from the other end. This continuous "on-power" fuel changing occurs while the reactor is operating rather than during shutdowns and results in a high net capacity factor, or energy delivered for a given amount of time divided by the energy if the reactor operated continuously at full power. [5,6] The low neutron absorption by the heavy water moderator results in a lower enrichment needed to burn the fuel, so low that uranium ore with natural fissile abundance of 0.72% can be burned in the reactor. This drops the fuel cost per unit energy produced even though the energy produced per unit fuel (fissile and fertile) mass in units of megawatt days per tonne (metric ton) of unenriched uranium (MWd/tonne U), or burnup, is around 7000 MWd/tonne U and lower than light water reactors around 30,000 MWd/tonne U. [4,5,7] Burnable poisons such as boron or gadolinium in the fuel deplete over time such that the reactivity remains constant as fuel depletes, and failsafes include 4 cadmium control rods as well as a backup injection of gadolinium nitrate into the heavy water moderator. [4]
There are some drawbacks associated with heavy water reactors that are absent in other designs. (1) The ability to burn uranium ore eliminates the need for enrichment and can therefore become a proliferation issue. (2) High-pressure piping is required unlike in molten salt reactor designs which, due to low vapor pressure of fluoride salts with higher melting points than water, can operate at atmospheric pressure. (3) While avoiding enrichment, the "once-through" fuel cycle without reprocessing is an inefficient use of the uranium supply even if it is currently more economically feasible. (4) In the current heavy water reactor design, fission products build up in fuel rod and eat the neutron population. This is more manageable in molten salt reactors where some neutron-eating fission products can be gassed or plated out and the fissile fuel concentration can be continuously adjusted.
The process of breeding refers to the maximization of the creation of fissile isotopes such as Pu-239 or U-233 from fertile U-238 or Th-232 absorbing a neutron and undergoing two beta decays. This can be accomplished in certain reactors designs by surrounding the reactor core with a blanket of fertile material which captures neutrons from burning fissile material. The process of breeding (albeit not efficiently) occurs in light water reactors since fissile material only makes up 3-5% of the fuel's uranium while the rest is neutron-absorbing fertile material. By the end of the fuel rod's life, about half of the energy is coming from the burning of plutonium created while burning the original U-235. We can characterize this process by defining the conversion ratio C as the ratio of the rate of new fissile isotopes creation (by fertile atoms absorbing neutrons) to the rate of fissile isotope (U or Pu) destruction while maintaining a critical reaction. [2,4] The equation for the conversion ratio is:
| C = ν - 1 - (A+L) | (4) |
where ν is the average number of neutrons produced in a fission event, A is non-productive absorption by fission products and other materials, L is leakage, and -1 is for the next fissile atom that maintains the chain reaction. C can therefore be thought of as the amount of "extra" neutrons left over after each fission that go towards breeding. Typically (A+L) ~ 0.3 depending on fuel and material choice. [2] When C > 1, it is known as the breeding ratio and the amount of fertile atoms made fissile relative to the natural fissile atoms consumed can be made arbitrarily large. In practice, this means more fissile fuel can be created than is being consumed while running the reactor. The conversion ratio C ~ 0.6 for light water reactors on enriched fuel, C ~ 0.8 for heavy water reactors on natural uranium, and C ~ 1.0 - 1.6 for liquid metal fast breeder reactors on higher enrichment. [4] The liquid metal fast breeder reactors do not use a moderator designed to thermalize the neutron population and instead use fast (MeV) neutrons created from fission events to (1) break down long-lived actinide waste and (2) be absorbed be fertile material. [1] The reactor is designed such that the neutron flux is increased over the epithermal region between eV and MeV energy scales; neutrons can then be absorbed in the cross section resonances of fertile fuel to breed fissile Pu-239 or U-233. Water is too effective at thermalizing the neutrons and thereby preventing significant absorption by fertile nuclei. Instead, fast breeder reactors use sodium (with potential fire risks) or lead-bismuth mixture as coolant.
More generally, the conversion ratio is defined as C = Rc / Xf where Rc is the integrated fertile capture rate and Xf is the integrated fissile capture rate (due to fissile nuclei both fissioning σf and absorbing σa). [8] The rate equation for neutron population criticality is given by: [4,8]
|
(5) |
The contributions to absorption are X = Xf + Rc + Xfp + Xcr + Xm where Xfp is the fission product absorption rate, Xcr is the control rod absorption rate, and Xm is the moderator absorption rate. Plugging the criticality equation into the conversion ratio equation yields:
|
(6) |
For C < 1, net fuel production is negative (i.e. fuel is consumed) and the neutron flux can be increased to maintain power by removing the control rods and reducing Xcr. [8] We can then identify the quantities in Eq. (4) with an effective neutron multiplication factor ν = G / Xf, absorption A = (Xfp + Xcr + Xm) / Xf, and leakage L = L / Xf. The rates are given by an integral over space and energy of the neutron flux: φ(r,E) = ρ(r)v(E)f(E) for neutron density ρ(r) with dimension [1/L3], neutron speed v(E) = (2E/m)1/2 with dimension [L/T], and neutron energy distribution f(E) with dimension [1/E]=[T2/ML2], multiplied by the macroscopic cross section: Σ(r,E) = n(r) σ(E) for isotope density n(r) with dimension [1/L3] and cross section σ(E) with dimension [L2] associated with the particular rate. [4,8] The dimension of flux is [L-2 T-1 E-1] and the dimension of macroscopic cross section is [1/L] such that the dimension of the rate is [(L3 E) (L-2 T-1 E-1) (L-1)] = [1/T]. The rates for fertile capture Rc, fissile generation G, fissile capture Xf, and moderator absorption Xm are given by [8]
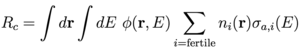 |
(7) |
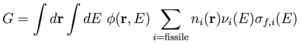 |
(8) |
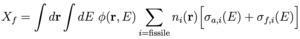 |
(9) |
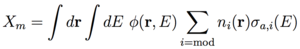 |
(10) |
For a heavy water reactor running on natural uranium, the parameters are neutron multiplication ν = 2.13, absorption A = (0.14 + 0.168) = 0.308 due to fission products and other materials respectively, and leakage L = 0.037 resulting in a conversion ratio C = 0.785. [2] To make thermal breeding possible, we can get a boost from increasing the average neutron multiplication factor or reducing non-productive absorptions/leakage. The neutron multiplication factor ν depends on the fissioning neutron's energy and the fissile material. For a fast neutron spectrum, Pu-239 and Pu-241 have the highest multiplication factor making the U-238/Pu-239 fuel cycle ideal for fast breeders reactors. [4] In contrast, U-233 has the highest multiplication factor ν ~ 2.3 at thermal energies making a Th-232/U-233 fuel cycle more suitable for maximizing neutron multiplication in heavy water reactors. [2,4] By additionally ignoring cost for the moment, the breeding ratio can be increased by up to ΔC ~ +0.125 through advanced techniques including improvement of isotope separation of light water component from heavy water coolant (from 99.75% purity to 99.95% purity) and use of on-power refuelling ability to temporarily remove fuel (allowing Pa-233 to decay into fissile U-233) before completing the burnup. [7]
We can compute a rough estimate of the conversion ratio in a heavy water reactor as a function of the fissile fuel enrichment ξ and fuel/moderator volume ratio Ω. The neutron flux φ(r,E) corresponds to a thermal spectrum (peaked around 0.026 eV) due to heavy water moderation. Let us make the simplifying assumption that the flux is independent of position r and the moderator concentration nm is constant in the moderating volume Vm and vanishes in the fuel volume Vf. Similarly the fissile U-233 and fertile Th-232 concentrations (with fissile enrichment ξ) are constants nfissile = ξ nf and nfertile = (1-ξ) nf in the fuel volume and vanish in the moderator volume. We can compute the rates G, Xf, and Xm:
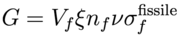 |
(11) |
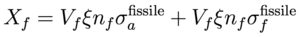 |
(12) |
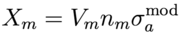 |
(13) |
where nf and Vf are the fuel concentration and volume respectively (and similarly for moderator nm and Vm), and constant ν is the thermally averaged neutron multiplication factor for fissile fuel. The fuel volume fraction Ω = Vf / (Vf + Vm) such that (1-Ω) = Vm / (Vf + Vm) and Vm / Vf = (1 - 1 / Ω). Using the criticality/breeding equations from the previous section and ignoring control rod loss (Xcr = 0) to maximize C, the conversion ratio is then given by:
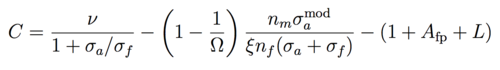 |
(14) |
We take the values for neutron multiplication ν = 2.3, fission product absorption Afp = Xfp / Xf = 0.083 and leakage L = L / Xf = 0.027 for Thorium equilibrium fuel cycle. [2] The density of the Th-232 is nf = 11.3 g/cm3 = 2.93 x 1022 cm-3. The fission cross section for U-233 is σf = 524 barns and absorption σa = 49. The heavy water moderator number density is nm = 2 x 3.68 x 1022 cm-3 and the absorption cross section σamod = 5.19 x 10-4 barns taken from Table 1. Plugging in the numbers, C ~ 0.993 over a range of ξ and Ω which suggests breeding is possible in heavy water reactors by incorporating some of the minor improvements of ΔC mentioned above.
Nuclear fuel is not renewable, and any reactor design hoping to serve as a baseload power supplier for the planet must be able to breed fissile fuel. While thermal heavy water reactors lack certain advantages over liquid metal fast breeders, they have a moderator evaporation failsafe present in light water reactors. By choosing a Th-232/U-233 fuel cycle to maximize the thermal neutron multiplication, a simple analysis suggests the better neutron economy of heavy water allows for a conversion ratio C > 1. Whether the necessary improvements to the conversion factor ΔC are cost-effective enough to actually implement must be further analyzed, but factoring in costs, a thorium equilibrium cycle should be feasible.
© John Dodaro. The author grants permission to copy, distribute and display this work in unaltered form, with attribution to the author, for noncommercial purposes only. All other rights, including commercial rights, are reserved to the author.
[1] R. Wilson, "The Changing Need for a Breeder Reactor," Nuclear Energy 39, 99 (2000).
[2] S. Banerjee, E. Critoph, and R. G. Hart, "Thorium as a Nuclear Fuel for CANDU Reactors," Canadian J. Chem. Eng. 53, 291 (1975).
[3] V. F. Sears, "Neutron Scattering Lengths and Cross Sections," Neutron News 3, No. 3, 26 (1992).
[4] W. M. Stacey, Nuclear Reactor Physics, 2nd Ed. (Wiley-VCH, 2007).
[5] J. A. L. Robertson, "The CANDU Reactor System: An Appropriate Technology," Science 199, 657 (1978).
[6] P. Bordia, "CANDU Reactors," Physics 241, Stanford University, Winter 2012.
[7] J. B. Slater, "An Overview of the Potential of the CANDU Reactor as a Thermal Breeder," Chalk River Nuclear Laboratories, AECL-5679, February 1977.
[8] D. R. Vondy, "On Nuclear Fuel, Mass Balances, Conversion Ratio, Doubling Time and Uncertainty," Oak Ridge National Laboratory, ORNL/TM-5050, November 1976.