 |
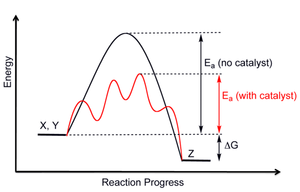
| Fig. 1: The free energy diagram of a chemical reaction with and without catalysts. (Source: Wikimedia Commons) |
The transformation and utilization of energy has always been one of the primary engines of the global economy since the industrial revolution, however, the challenges of a changing planet, rising commodity costs, and a greater awareness of the environmental impact of traditional forms of energy has lead to a focus on new energy resources such as natural gas and other renewable energy source. However, one of the biggest problems with renewable energy has been in the storage and transportation of energy. [1] While the breakthroughs in energy recovery and renewable energy generation has lead to an abundant source of natural gas as well as the promise of greenhouse gas free electricity, much of our existing technology and infrastructure cannot easily utilize these energy sources. In order to convert, store, and utilize the energy from these new sources, we need catalysts. [2,3]
Catalysts, in the broadest definition, is a material that increases the rate of chemical reaction. They accomplish this task by lowering the transition barrier of the intermediate state between the reactant and the final product, as shown in Fig. 1. While they do not change the overall enthalpy of a chemical reaction, they can greatly influence the kinetics, or the speed, or a chemical reaction. [4] Why do we care about kinetics? Because in order for us to transform sufficient quantities of material from one chemical to another chemical, it has to be fast and cheap.
First, the process must be fast so that it can occur at production speed. Perhaps one of the most extreme example of this diamond and graphite. The chemical reaction going from Diamond to Graphite is energetically favorable. However, because of the kinetics of the chemical reaction from Diamond to Graphite under our atmospheric conditions are so slow, this process is too slow to be observed. Only under highly favorable conditions for that reaction, can we observe the this transformation in a reasonable time-frame. [5]
 |
| Fig. 2: This is a fluidized catalytic cracking reactor in Texas. (Source: Wikimedia Commons) |
Then, the process much be cheap. Ideally, this means that the target reaction can occur at a sufficient rate without extreme energy differences driving the reaction. Take the example of the classic Fischer-Tropsch process of gas to liquid fuels. A novel carbon- nanotube encapsulated Iron catalyst has 6 times higher specific activity than traditional active carbon supported iron catalysts towards C5+ hydrocarbons. [6] This leads to an increased efficiency in producing the desired product, which also means it is cheaper and less energy intensive to create the product.
Catalysts are already shaping our energy consumption and usage. Since the early days of the petroleum industry, Catalysts have been used to convert less valuable crude oil products, such as asphalt and tar, to shorter chain product like diesel, gasoline, and kerosene. This process called cracking, which is conducted in large chemical process facilities such as the one shown in Fig. 2, has been increasing the ability for us to recover more value-added products from crude oil. [7]
The increased abundance of natural gas due to the fracking boom in the United States has also lead to a resurgence in the investigation of gas to liquid technologies including novel catalysts. It is estimated that 140-170 billion cubic meters of natural gas are flared off every year in the recovery process of fossil fuels. [8] Although natural gas is readily used as a fuel source, their lackluster energy volumetric density as well as our lack of infrastructural support for using natural gas as a transportation fuel has lead to a lack of utilization. If we can effectively convert natural gas to conventional liquid fuels, that can greatly reduce the amount of greenhouse gasses emitted into the atmosphere by reducing all the wasted flares, and thus displacing a fraction of the need to increase oil production. [9]
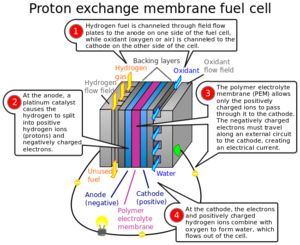 |
| Fig. 3: A diagram of a hydrogen fuel cell labeled with the role of the platinum catalyst at the anode of the fuel cell. (Source: Wikimedia Commons) |
Catalysts are also critical in our goal of increasing the adoption of renewable energy sources. One such source is the conversion of plant biomass into carbon-neutral liquid fuels. The process uses the same technology as the traditional Fischer-Tropsch process, however due impure chemical nature of plant matter, the catalysts used in this process must be more resistant to catalytic poisoning, a process where undesired reactants react with the catalyst in such a way that it decreases its effectively towards the desired product. [10]
The solar industry is also looking into using catalysts as a means of energy storage. Solar is highly dependent on daylight and weather, thus one of the biggest challenges to the implementation of solar is the storage problem. [11] Water splitting catalysts presents an interesting potential solution to that problem. Using just water, the excess electricity produced by solar can be used to split water into hydrogen and oxygen, which can be then recombined at a later time in a fuel cell in order to produce electricity. The Catalyst in that case is crucial in reducing the overpotential needed to drive the kinetics of the water splitting reaction reaction, thus decreasing conversion losses in energy. [12]
The catalyst is also crucial in unlocking fuel cells as a commercially viable method of generating electricity. Currently, the conversion between hydrogen and oxygen is mediated with expensive platinum and other noble metal catalysts that are prohibitively expensive. [13] The predominant catalyst for the half-reaction at the anode shown in Fig. 3 is predominantly platinum, which is extremely cost prohibitive. New research in that area has produced some promising new catalysts using iron and manganese that are cheaper to produce. [14]
The rapidly increasing change in our energy consumption profile combined with our existing infrastructure necessitates that we transform the energy into materials that we can readily use. The demand for energy dense, easily used, and cheap fuel has never been higher. The field of catalytic research is poised to make multiple breakthroughs in both traditional and renewable energy fields.
© Peter Wang. The author grants permission to copy, distribute and display this work in unaltered form, with attribution to the author, for noncommercial purposes only. All other rights, including commercial rights, are reserved to the author.
[1] M. M. Thackeray, C. Wolverton, and E. D. Isaacs, "Electrical Energy Storage For Transportation - Approaching the Limits of, and Going Beyond, Lithium-Ion Batteries," Energy Environ. Sci. 5, 7854 (2012).
[2] G. A. Olah, "Beyond Oil and Gas: The Methanol Economy," Angew. Chem. Int. Ed. 44, 2636 (2005).
[3] M. Krumpelt et al., "Fuel Processing For Fuel Cell Systems in Transportation and Portable Power Applications," Catal. Today 77, 3 (2002).
[4] M. Boudart and G. Djéga-Mariadassou, Kinetics of Heterogeneous Catalytic Reactions (Princeton University Press, 2014).
[5] Y. G. Gogotsi, A. Kailer, and K. G. Nickel, "Materials: Transformation of Diamond to Graphite," Nature 401, 663 (1999).
[6] W. Chen et al., "Effect of Confinement in Carbon Nanotubes on the Activity of Fischer-Tropsch Iron Catalyst," J. Am. Chem. Soc. 130, 9414 (2008).
[7] J. Ancheyta, S. Sánchez, and M. A. Rodríguez, "Kinetic Modeling of Hydrocracking of Heavy Oil Fractions: A Review," Catal. Today 109, 76 (2005).
[8] C. D. Elvidge et al., "A Fifteen Year Record of Global Natural Gas Flaring Derived From Satellite Data," Energies 2, 595 (2009).
[9] A. Y. Khodakov, W. Chu, and P. Fongarland, "Advances in the Development of Novel Cobalt Fischer-Tropsch Catalysts For Synthesis of Long-Chain Hydrocarbons and Clean Fuels," Chem. Rev. 107, 1692 (2007).
[10] G. W. Huber, S. Iborra, and A. Corma, "Synthesis of Transportation Fuels From Biomass: Chemistry, Catalysts, and Engineering," Chem. Rev. 106, 4044 (2006).
[11] N. S. Lewis and D. G. Nocera, "Powering the Planet: Chemical Challenges in Solar Energy Utilization," Proc. Natl. Acad. Sci. (USA) 103, 15729 (2006).
[12] P. Du and R. Eisenberg, "Catalysts Made of Earth-Abundant Elements (Co, Ni, Fe) For Water Splitting: Recent Progress and Future Challenges," Energy Environ. Sci. 5, 6012 (2012).
[13] A. F. Ghenciu, "Review of Fuel Processing Catalysts For Hydrogen Production in PEM Fuel Cell Systems," Curr. Opin. Solid St. M. 6, 389 (2002).
[14] R. Burkitt, T. R. Whiffen, and E. H. Yu, "Iron Phthalocyanine and MnOx Composite Catalysts For Microbial Fuel Cell Applications," Appl. Catal. B-Environ. 181, 279 (2016).