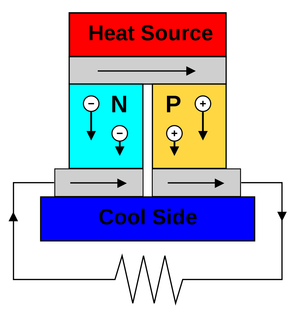
 |
| Fig. 1: Schematic diagram of a thermionic electricity generator as might be used to make power from human body. (Source: Wikimedia Commons) |
The idea of converting human body heat into a form of usable energy has been targeted by scientists for years. A resting human male gives off roughly 100-120 Watts of energy. A very small fraction of this can be utilized by a thermoelectric device to power wearable devices. As an example, Seiko's Thermic Watch managed to capture continuous a charge at only one microwatt, so there is certainly a viable market that has not yet blossomed. It being known that 80% of a typical human's body power is given off as heat, current technologies can only produce a few milliwatts of energy. This is not nearly enough to charge your iPhone at a power input of roughly 5 watts, and unfortunately this sort of efficiency may never be possible.
Because we must keep in mind energy cannot be conserved absolutely, we must look to the origin of this energy in the means of food, and calories in particular, that the human body consumes as the source of this heat energy through metabolism. A single "calorie" of food contains 4.184 × 103 joules. If the average human consumes 1500 calories each day, that translates to 6.27 × 106 joules per day. Relatively speaking, this is about the amount of energy required to run a car for 15 minutes. On a global scale, this translates to roughly 3.14 × 1019 J per year. [1] Having this amount of energy input in mind, we also need to understand the entropy involved The second law of thermodynamics states that no system is entirely efficient, and some of this energy must be given off as waste heat, or body heat as we know it.
Current thermoelectric energy conversion is completed largely through the use of the Seebeck Effect. Fig. 1 provides a simple diagram displaying the functional mechanism. Thermoelectric devices generate energy when there is a difference between the heated surface (in contact with the body) and the environment. This can be written in the form dT/T, where dT is the difference in temperature between body and the environment, where T is the temperature of the environment in Kelvin. If the human body has a temperature of 310°K (98.6°F), and the environment has a temperature of 297°K (75°F), we get a dT value of 13°K over a T value of 310°K. This equals a value of 4.2% in available differential. This value will actually be much lower given the inability to utilize all heat lost, as this would be fatal for any living human. [2] Qualitatively speaking, more electricity would be generated by a device worn by a user in a colder environment. In function, electrons travel from areas of high temperature to areas of low temperature. In order to harness the current, two semiconductors are necessary. One n-type (negative) and one p-type (positive) that form a thermoelectric couple to ensure there is a continuous current with which power can be produced, as can be seen in Fig. 2. They must be configured in series electrically but thermally in parallel. [2] The larger the differential in temperature, the larger the corresponding voltage will be, and the larger the ability of the current to power a load (i.e. your Apple Watch), effectively converting thermal energy to electrical energy.
More robust thermoelectric generators (TEG) that are used in larger industrial applications like gas pipelines, steel foundries, and combined heat and power plants, have the ability to generate much larger heat difference and thus a much larger voltage. [3] Based on their larger size and available space, many units can be linked in series, driving a larger power output. Thermoelectric devices harvesting body heat, however, must be much smaller, much more flexible, and can't drive as large of a temperature difference. These factors result in a much lower power output in the range of micro to milliwatts. These limitations aside, creating a larger figure of merit, or Z value, which is the ratio of efficiency between the thermal properties and electrical properties of the materials used, drives a larger power output. [4] Advances in nanotechnology have been made to manipulate the structure of materials to create a higher Z value, although these processes are extremely complex and there are limitations to how much increase in Z value these can create.
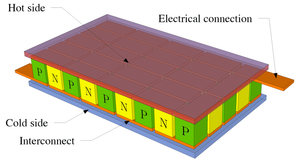 |
| Fig. 2: A thermoelectric device configuration (Source: Wikimedia Commons) |
Our main consideration in developing materials with as high of a Z value as possible is finding out how to reduce the thermal conductivity. [5] This is where nanotechnology comes into play. By using carbon nanotubes and graphene, researchers can develop nanostructure smaller than the wavelength of light. This means that there is an enhancement in the scattering of photons, which results in a decrease in thermal conductivity. [5] With the electrical conductivity still in place, the Z value becomes greater, making the material more effective in a thermoelectric generator. This is particularly valuable in harvesting body heat, as a higher Z value for flexible nano materials allows a much higher conversion of body heat to electrical energy. Thus, these devices can compensate, in a minor way, for the smaller differential in temperature in producing a larger voltage. At the moment, these nanostructure have not been efficient enough to be produced commercially. With further research we can expect this to change to some degree.
On a wider scale, thermoelectric devices allow us to capture wasted energy through heat, which is often a byproduct of other energy producing methods. In the wearable market, this means that we could harness our body's heat energy to provide unlimited power to certain small devices as long as the environment is cold enough. However, there is an extremely marginal value of power output that can actually be realized, and this is driven by three factors. First, the amount of heat actually lost through the human body. Second, the ability to drive a large enough dT/T. Lastly, the ability to actually harness this dT/T, as we can only safely and practically capture a small percentage of human body heat. Thus, the overall power output we could project to be harnessed by the entire human race (if everyone were to possess a device), wouldn't even put a dent in the overall global energy requirements. Thus, as a large scale energy solution, it is not feasible, but as a niche application, it is intriguing.
© Matthew Stevens. The author grants permission to copy, distribute and display this work in unaltered form, with attribution to the author, for noncommercial purposes only. All other rights, including commercial rights, are reserved to the author.
[1] D. Bechstein, "World Food Budget," Physics 240, Stanford University, Fall 2013.
[2] M. Highland eet al., "Wearable Thermoelectric Generators for Human Body Heat Harvesting," Appl. Energy 182, 518 (2016).
[3] F. DiSalvo, "Thermoelectric Cooling and Power Generation," Science, 285, 5428 (1999).
[4] V. Leonov, "Thermoelectric Energy Harvesting of Human Body Heat for Wearable Sensors," IEEE Sens. J. 13, 2284 (2013).
[5] G. Pennelli et al., "Review of Nanostructured Devices for Thermoelectric Applications," Beilstein J. of Nanotechnology, 5, 1268-1284, (2014).