 |
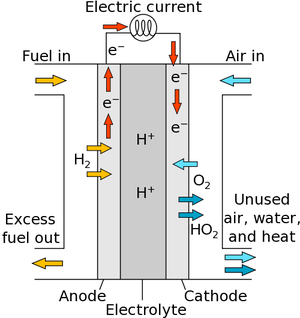
| Fig. 1: Diagram of Fuel Cell Operation. (Source: Wikimedia Commons) |
Fuel cells are a technology that can generate electricity through a chemical reaction process that utilizes a fuel source and oxygen gas. There are three components to a fuel cell: the anode, the cathode, and an electrolyte (see Fig. 1). The fuel enters the anode side of the fuel cell, while oxygen enters the cathode side. A catalyst in the anode then assists in splitting the molecules of the fuel into electrons and positively charged ions. These charged ions are then drawn though the electrolyte layer between the anode and cathode. As the electrolyte layer is designed to only allow positively charged particles from passing through, the negatively charged electrons are forced to flow from the anode to the cathode via an external circuit, which then creates an electric current. When the charged ions and electrons reach the cathode, they react with oxygen gas to form a new molecule, a byproduct of the reaction. For the case of hydrogen fuel cells, water is the byproduct created.
In the realm of automobiles, there have been concerns over the decreasing reserves of oil worldwide and the increasing role of conventional petroleum driven vehicles as a major contributor to environmental issues. So, there has been a need for the automotive industry to develop vehicles that can utilize alternative fuel sources. The most popular type of alternative fuel vehicles is the battery electric vehicle (BEV), which already have many successful models out on the road (such as the Nissan Leaf, Tesla Model S, Volkswagen E-Golf, BMW i3, etc). A BEV stores energy on a large onboard battery, which drives an electric motor to propel the vehicle. The hydrogen fuel cell vehicle (HFCV) is another growing category of alternative fuel vehicles. Similar to BEVs, HFCVs also utilize electric motors for propulsion. However, instead of batteries, the fuel cells onboard generate the required electricity by consuming compressed hydrogen gas. Single fuel cells are only capable of producing small electrical potentials. So, for this technology to be effective for automobiles, numerous fuel cells are connected together in series, called a stack. There has been very limited commercial production of HFCVs. However, there have been increased efforts to improve market share in recent years with the advent of the Toyota Mirai (see Fig. 2) and the Honda Clarity Fuel Cell.
 |
| Fig. 2: Toyota Mirai at ITM Hydrogen Refueling Station. (Source: Wikimedia Commons) |
To examine the viability of hydrogen fuel cell vehicles, it is necessary to compare HFCVs with BEVs, the competitor in the area of alternative fuel vehicles. One traditional issue with battery electric vehicles is the driving range. Most BEVs are capable of travelling only short distances. The Nissan Leaf, for example, only has an effective range of 100 miles. [1] Also, only the high end battery electric vehicles, such as Tesla Model S, can achieve range comparable to conventional petroleum driven vehicles (294 miles). Hydrogen fuel cell vehicles, on the other hand, do not appear to suffer from these limitations. It is reported that the Toyota Mirai can travel up to 312 miles before refueling is needed. The Honda Clarity Fuel Cell has even greater range at 366 miles. [2]
The amount of time needed to refuel is another area that hydrogen fuel cell vehicles excel at. The time it takes for HFCVs to refuel is only 3 to 5 minutes, which is comparable to that of conventional internal combustion vehicles. [3] This is significantly much better than battery electric vehicles, which at best will require more than 30 minutes of charging time to regain full range. [4] So, from a user experience perspective, hydrogen fuel cell vehicles may be preferable to battery electric vehicles.
The environmental considerations of hydrogen fuel cell vehicles are also important. There have been studies that have compared HFCVs with other types of existing vehicles (conventional, hybrid, and battery electric). [5] In terms of the creation and utilization of fuel, HFCVs appear to create less greenhouse gas (45%) and air pollution (70%) emissions compared to conventional vehicles. Depending on the energy generation process, HFCVs also appear to be comparable to BEVs.
However, hydrogen fuel cell technology also has some major drawbacks. The data suggests that HFCVs may not be as environmentally friendly as we think. The production of HFCVs generates a significantly greater amount of greenhouse gas emissions and more than twice as much as the other types of vehicles. [5] A similar trend can be seen for air pollution emissions. These HFCVs cause more than 4 times air pollution emissions as much as compared to conventional vehicles. Hydrogen fuel cell vehicles also appear to be less efficient at utilizing electricity (19-23%) compared to battery electric vehicles (67%). [6] In BEVs, electricity only needs to be transmitted from the energy grid to charge the vehicle's battery. However, with HFCVs, electricity is first used to create hydrogen gas through electrolysis. Afterwards, the hydrogen undergoes compression or liquefaction so that it can be better transported. Lastly, the hydrogen needs to be consumed by the fuel cells before electricity can be used. As there are significant energy losses with each of these additional steps for HFCVs, it is clear why there is such a large discrepancy in efficiency at utilizing electricity between HFCVs and BEVs. So, while hydrogen fuel cell vehicles are lauded as a "green" zero emissions technology, there are some hidden costs associated with the production of HFCVs.
From a practicality perspective, hydrogen fuel cell vehicles have encountered some challenges. In terms of time required to refuel, HFCVs are comparable with the conventional petroleum driven vehicles and are much better than BEVs. However, users of HFCVs often lack the flexibility as to where they can refuel. There is a minimal amount of supporting infrastructure available to drivers of hydrogen fuel cell vehicles, with only 34 public fueling stations in the United States, with 31 of the said 34 stations being located in California. In comparison to battery electric vehicles, there are 15,703 publicly accessible electric charging stations for BEVs around the United States. [2] This lack of fueling stations for HFCVs is a major inconvenience to drivers and makes it impractical to own a hydrogen fuel cell vehicle outside of California. Furthermore, as there are so few HFCVs on the road and each fueling station is estimated to cost 2 million dollars, most companies are not motivated to construct additional infrastructure and fueling stations until it is clear that hydrogen fuel cell vehicles are viable. [7] Comparing the cost with other vehicles, HFCVs are also much higher than the conventional vehicles and BEVs. However, the prices of hydrogen fuel cell vehicles may become more competitive in the future. [8]
Hydrogen fuel cell vehicles fill an important niche in the spectrum of alternative fuel solutions. While HFCVs do have some advantages / benefits over the other competitors (such as BEVs), they have also encountered many challenges. If the construction of supporting infrastructure and fueling stations continue to stagnate, hydrogen fuel cell vehicles may have difficulty in increasing its market share among competitors.
© Brian Mok. The author grants permission to copy, distribute and display this work in unaltered form, with attribution to the author, for noncommercial purposes only. All other rights, including commercial rights, are reserved to the author.
[1] C. Fleming, "Chevy Bolt EV Range is 238 miles: Prime Time For the Electric Car?," Los Angeles Times, 12 Sep 16.
[2] "Hydrogen Fuel Cell Cars Creep Up - Slowly - on Electric Vehicles," Los Angeles Times, 13 Apr 17.
[3] C. Fleming, "2017 Honda Clarity: A Hydrogen Fuel Cell Car That Delivers Fun," Los Angeles Times, 30 Mar 16.
[4] C. Fleming, "How Will I Charge My Electric Vehicle? And Where? And How Much Will It Cost?," Los Angeles Times, 26 Sep 16.
[5] M. Granovskii, I. Dincer, and M. A. Rosen, "Economic and Environmental Comparison of Conventional, Hybrid, Electric and Hydrogen Fuel Cell Vehicles," J. Power Sources 159, 1186 (2006).
[6] U. Bossel, "Does a Hydrogen Economy Make Sense?" Proc. IEEE 94, 1826 (2006).
[7] D.-A. Durbin, "Hydrogen Fuel Cell Cars Face Obstacle: Few Fueling Stations," Jan Jose Mercury News, 14 Apr 17.
[8] G. J. Offer et al., "Comparative Analysis of Battery Electric, Hydrogen Fuel Cell and Hybrid Vehicles in a Future Sustainable Road Transport System," Energy Policy 38, 24 2010).