 |
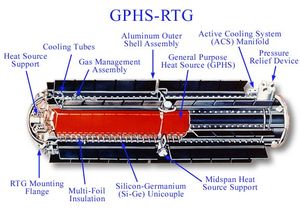
| Fig. 1: A diagram of an RTG. (Source: Wikimedia Commons) |
Few Americans are aware of nuclear energy's dominant position as the primary power source for U.S. activities in space. Nuclear energy has been used to power U.S. spacecraft since 1961, when the U.S. Navy first used a radioisotope thermoelectric generator (RTG) to power the Transit 4A navigation satellite. At present, RTGs can be found in the furthest man-made objects from Earth, Voyagers 1 and 2. An RTG is well suited to spacecraft that will travel too far from the sun (or on the dark side of planets) to rely upon solar power. [1] See Fig. 1 for a diagram of a typical RTG.
An RTG is distinct from a nuclear reactor, which generates energy by splitting atoms. It converts the heat expended by the decay of plutonium-238 (Pu-238) into electricity using thermocouples--devices that convert thermal energy into electrical energy. [2] A General Purpose Heat Source (GPHS) module, which contains 600 grams of Plutonium dioxide that produces 250 watts of thermal power, supplies the converter with heat. Some heat is converted into electricity, while the rest is expelled as waste heat that is subsequently used to warm the spacecraft's instruments. [1]
What is notable about the RTG is its ability to convert heat into useful electricity without moving parts. The lack of moving parts simplifies operation of the power sources in space, whereas traditional engines used on Earth's surface are more likely to malfunction outside of Earth's atmosphere.
 |
| Fig. 2: An example of a Pu-238 pellet. (Source: Wikimedia Commons) |
RTGs use Pu-238. The isotope's half-life of 88 years and stability at high temperatures make it ideal for the conditions under which an RTG must operate. Additionally, Pu-238 requires minimal shielding equivalent to that of a piece of paper, and its low levels of radioactivity mean that instruments and equipment are unaffected.
The plutonium itself takes a ceramic form (see Fig. 2) and is broken into chunks of solid material. The solid state is preferable to a gaseous one as it reduces the risk of harm to humans and the environment in the event of an accident. In the 50 years since the RTG's introduction, every RTG has functioned safely and as intended.
Pu-238's emissions consist of alpha particles. In the context of Pu-238 emissions, alpha particles do not harm human tissue, as they do not penetrate outer layers of skin cells. [3] (Some alpha particles, such as those present in cosmic rays, possess enough energy to penetrate live tissue, as well as thin layers of metal. However, these high-energy alpha particles are not pertinent to this discussion.)
The primary concern regarding Pu-238 is its limited supply. In contrast to Pu-239, Pu-238 is not weapons grade and therefore has not been produced in great quantities. When the U.S. Department of Energy shut down its reactors in the late 1980s, domestic Pu-238 production halted. The U.S. supplemented its dwindling inventories by purchasing Pu-238 from Russia, who soon ended production, as well. [4] Currently, there is enough Pu-238 to last through 2022. If the U.S. does not resume production of Pu-238, NASA will not be able to explore the solar system beyond Mars. [5]
© Alexandra Crerend. The author grants permission to copy, distribute and display this work in unaltered form, with attribution to the author, for noncommercial purposes only. All other rights, including commercial rights, are reserved to the author.
[1] D. J. Anderson, "NASA Radioisotope Power Conversion Technology NRA Overview," U.S. National Aeronautics and Space Administration, NASA/TM-2005-213981, November 2005.
[2] D. Kramer, "Shortage of Plutonium-238 Jeopardizes NASA's Planetary Science Missions," Physics Today 64, No. 1, p. 24 (2011).
[3] R. C. O'Brien et al., "Safe Radioisotope Thermoelectric Generators and Heat Sources for Space Applications," J. Nucl. Mater. 377, 506 (2008).
[4] D. Kramer, "Shortage of Plutonium-238 Jeopardizes NASA's Planetary Science Missions," Physics Today 64, No. 1, 24 (2011).
[5] A. Lawler, "Technology is Essential, But It's a Tough Sell," Science 295, 39 (2002).