 |
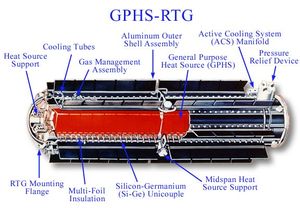
| Fig. 1: Diagram of an RTG used on the Cassini probe. (Source: Wikimedia Commons) |
Reliable, long-lasting batteries are crucial for any item that needs a power source, like cell phones, laptops, cars, and flashlights. Batteries' role in everyday life is important, and a large body of research seeks ways to improve battery capacity, efficiency, and life. As the threat of climate change and energy insecurity loom, nuclear options emerge.
Nuclear batteries, which use energy from the decay of radioactive isotopes to generate electricity, are attractive despite their cost because they have the potential for a very long battery lifetime (10-20 years), longer shelf-life, and higher energy density, compared with other energy storage methods. [1] Although they use atomic energy to generate electricity, nuclear batteries differ from nuclear reactors because no chain reactions are utilized. Nuclear batteries mainly serve as power sources for equipment that requires long operation in remote places, including medical implants like pacemakers [2], spacecraft, or remote military sensing, particularly before lithium-ion batteries were prevalent. However, several challenges face the miniaturization of nuclear batteries.
Prelas et al. published a review of efforts to miniaturize nuclear battery systems that is briefly covered here. [1] The review is not optimistic, stating that the physics of nuclear batteries do not currently support the objectives of miniaturization, high efficiency and high power density. Instead, the physics imply that nuclear batteries will be of moderate size and limited power density because the two properties desired most of a nuclear battery (long shelf- life and high power density) are opposed because of the fundamentals of nuclear decay: energy density varies inversely with isotope half-life. [1]
The performance of nuclear batteries is ultimately a function of the radioisotope(s), radiation transport, and energy conversion transducers; these vary significantly among nuclear battery types. [1] The energy conversion efficiency of non-thermal-type nuclear batteries demands that the system's two governing length scales (the ionizing radiation range and the transducer size) be approximately equal. [1] Mismatches are the limiting factor in theoretical energy conversion efficiency of small-scale nuclear batteries. [1] Power density is dictated by the transducer- radioisotope interface; solid radioisotopes typically are coated on the transducer and thus scale power density with surface area, limiting performance. [1] However, if isotopes are embedded within the transducer, the power density can scale with cell volume and maximizes the power density. [1] Previous work has demonstrated that liquids are effective shielding materials for beta radiation and can help provide this higher power density.
Various nuclear battery types exist, from thermoelectrics to betavoltaics, and have been studied since the 1950s. Prior art has predominantly investigated solid-state materials, but existing designs are limited by performance degradation as a result of radiation damage to semiconductors [2].
Radioisotope Thermoelectric Generator (RTG): This is the dominant nuclear battery technology. (See Fig. 1.) It is considered the performance standard for all nuclear battery types and has been used in many deep space missions. [1] It operates by the Seebeck effect; the enormous size of RTGs ensures that all of the radioisotope energy is deposited in the transducer and converted into heat. [1] Unfortunately, RTGs have poor efficiency (6%) and are very big: their low power density makes them poor candidates for downscaling. [1]
Betavoltaic Cells: These are also common, but studies have shown that only a small fraction of the entire radiation energy can be converted into useful electrical energy, and their lattice structures are prone to radiation damage from the high-energy beta particles, which causes consequent performance degradation. [2,3]
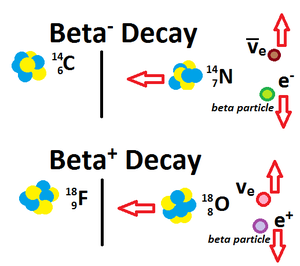 |
| Fig. 2: Diagram of beta decay. |
Recently, a team led by associate professor of electrical and computer engineering and nuclear engineering Jae W. Kwon at the University of Missouri published ground-breaking work in Nature on the development of a novel water-based radiolytic cell powered by beta radiation. [2] One of the major contributions of their design is the use of a liquid-phase medium to efficiently absorb the beta particles' kinetic energy, a main shortcoming of the betavoltaic cell. [2] Kwon's team says that the importance of this work lies largely in the new mechanisms for power generation in radiolytic chemical cells and may enable future work toward producing chemical cells with impressive power density.
Although not the first nuclear battery ever created, their work represents the first nuclear battery ever to use radiolysis to generate electric current at lower temperatures and higher energy levels than was thought possible, especially compared with other water-splitting energy production technologies. The beta radiation also continuously generates free radicals in liquid at room temperature and pressure, which can in can also generate electrical energy. [2] Other purported benefits include long-life and high efficiency (η = 53.88% at a potential of -0.9V at the platinum/electrolyte interface). [2]
It should be noted that this efficiency is defined as η = Pout / Prad , where Pout is the output power density of the device and Prad is the radiation power density of the source. [2] To calculate Prad, the average kinetic energy of Sr-90/Y-90 (490.96 keV) is used because this was the source of the beta particles in Kwon's experiments. Sr-90 is a highly dangerous substance that is unlikely to be used near people in commercial applications of the battery.
The water splitter used in Kwon's work is a platinum-coated, nanostructured semiconductor; its nanoporous surface permits far more chemical reaction sites than a flat one would. [2] This nanostructure is an electrode that acts as a catalyst for water decomposition and is made of titanium dioxide (TiO2). TiO2 is a material that is common and stable and has a large band gap (3.2 eV) that can improve radiolysis yield. [2] TiO2 is also very resistant to corrosion, necessary here because high pH values are required for water splitting (1 M KOH aqueous solution was used). [2]
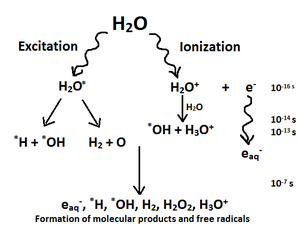 |
| Fig. 3: Diagram of water radiolysis. |
As high-energy beta radiation passes through the Pt and TiO2, the TiO2 nanostructure produces electron-hole pairs, which creates an electron flow and resultant electric current. Beta particles are high-energy, high-speed electrons or positrons that are generated when the neutron to proton ratio within the nucleus is unstable because it is too large (see Fig. 2). [2] The holes move toward the Pt/liquid interface while the electrons move to the other electric contact due to electric potential at the Pt/electrolyte interface. [2] As in nuclear reactors, the battery cell has a water-based semiconductor lattice structure that acts as a buffer by providing shielding from the radioactive source and absorbing large amounts of beta particles' kinetic energy, or nuclear energy. This energy absorption/loss of kinetic energy protects the semiconductor and also generates free radicals. [2]
As this radiation energy is efficiently absorbed, water radiolysis occurs. As high-energy electrons pass through an aqueous solution, they can ionize or excite water molecules. Radiolysis in general involves the dissociation of molecules via nuclear radiation; water radiolysis is the splitting of water molecules to produce free radicals that are powerful reducing and oxidizing (redox) agents (see Fig. 3). [2] Free radicals are a highly reactive but short-lived chemical species that can also be converted into electricity. As mentioned above, TiO2 has a large band gap. This permits beta particles' ionization energy to excite electrons while remaining far below the TiO2 band gap, thus making the beta particles a reliable electrical energy source that can expand the cell's power output and provide the impressively high output power density under irradiation. [2]
Speculations on eventual use of the new technology range from emergency applications and car batteries to complex, energy-hungry applications like spacecraft because the ionic solution does not freeze easily at quite low temperatures and the battery does not require recharging. [2] Although some may be leery of using nuclear energy in consumer products, Kwon says betavoltaics and other controlled nuclear technologies are not inherently dangerous, citing existing commonplace commercial applications including bedroom fire detectors or emergency exit signs in buildings. [2]
© Natalie Burkhard. The author grants permission to copy, distribute and display this work in unaltered form, with attribution to the author, for noncommercial purposes only. All other rights, including commercial rights, are reserved to the author.
[1] M. A. Prelas et al., "A Review of Nuclear Batteries," Prog. Nucl. Energ. 75, 117 (2014).
[2] B. H. Kim and J. W. Kwon, "Plasmon-Assisted Radiolytic Energy Conversion in Aqueous Solutions," Nature Sci. Rep. 4, 5249 (2014).
[3] S. Harrison, "Betavoltaic Devices," PH241, Stanford University, Winter 2013.