 |
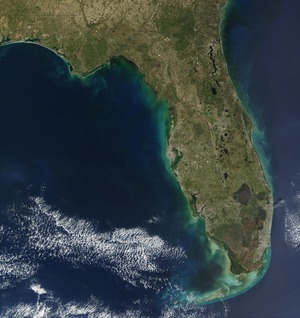
| Fig. 1: Red Tide Bloom. (Acquired by the Moderate-Resolution Imaging Spectroradiometer (MODIS) on NASA's Terra satellite, 17 Feb 02. Courtesy of NASA.) |
Nitrogen, taking up 79% of the atmosphere with the form of N2, is one of the major components that is necessary for life. Unfortunately, with most of it in the unreactive form of N2 gas, the reactive form of nitrogen is insufficient to sustain the human life on earth. [1] Human then started to intervene and developed technologies to convert N2 to reactive nitrogen compounds, in order to enrich the soil that the crops grow on.
However, the amount of reactive nitrogen produced is soaring and the nitrogen pollution is distributed globally, not just by wind and water naturally, but also by international commerce, like ships and trucks. [2] Up until now, the nitrogen cycle is seriously out of control by the perturbation from human action.
The nitrogen cycle consists of four processes: denitrification. [3] All organisms need to wait for nitrogen to be "fixed" by the plants before they can extract the nutrients out of NH4+, NO3- , the main compounds that they can use. [4] The two major natural sources of new nitrogen entering this cycle are lightening and nitrogen-fixing organisms, a relatively small number of algae and bacteria. [4]
The standard unit of measuring nitrogen cycle is teragram (Tg), which is equivalent to a million tonnes of nitrogen. The combination of the biologically available nitrogen fixed by lightening (5~10 million tonnes) and microbes (120~140 million tonnes) on land is around 140 million tonnes. [4] In the nitrification process, the nitrate, which is the most important form of nitrogen in terms of agriculture, is formed. [4] In the process of denitrification, nitrite and nitrate is reduced to nitrogen oxides and molecular nitrogen. Table 1 illustrates the nitrogen cycle.
The biggest human source of reactive nitrogen comes from nitrogen fertilizer. Currently, the Haber-Bosch process is used to produce about 100 Tg of reactive nitrogen per year worldwide and the global use of nitrogen fertilizers is increasing by about 15 Tg per year. [2] Compared to natural source of reactive nitrogen of 140 Tg annually, the human source is considerably altering the nitrogen cycle.
|
||||||||||
| Table 1: Nitrogen Cycle. [3] |
Excess nitrogen causes numerous health and environmental problems listed below: [5]
Coastal dead zones and fish kills due to severe eutrophication or hypoxia resulting from nitrate run-off and leaching into river systems. Although the red tide in Fig. 1 isn't caused by man-made nutrient source, but it is capable of using man-made nutrients for growth.
Biodiversity loss in terrestrial, freshwater and coastal water systems due to eutrophication and acidification.
Groundwater pollution by nitrates.
Freshwater pollution due to eutrophication and acidification.
Human health impacts resulting from the formation of aerosols and ground-level (tropospheric) ozone, a main component of smog, causing respiratory diseases.
Reduced crop, forest and grassland productivity due to nitrogen deposition and over- fertilization, as well as ground-level ozone exposure.
Global climate change and the depletion of stratospheric ozone, which protects life on Earth from harmful ultraviolet (UV) rays.
Impractically speaking, if many of the world's meat-eaters were to switch to a largely vegetarian diet, farmers could plant far less nitrogen-stoked grain, most of which goes to animal feed and sweeteners. [2] But meat consumption in the United States and Asia is rising rather than falling. [2]
A more practical, low-tech, low-cost solution is to improve the ways farmers rotate crops and fertilize their lands. Instead of adding fertilizer in fall, as most of the farmers in the American Midwest do, the alternative is to add exactly the right amount of fertilizer exactly when it is needed, preventing a big portion of the fertilizer from being washed away by winter snow and spring thaw. [2]
Other policies suggest that focusing on natural production (nitrogen "fixing") while limiting flows of reactive nitrogen and fixing nitrogen in such a way that it does not find its way back into the environment might be a useful solution. [5] Moreover, reusing nitrogen and using technology that converts nitrogen back into unreactive nitrogen (N2) as soon as possible would be a great solution to control the excess amount of nitrogen. [5]
Nitrogen is vital as an energy supply to grow food for the soaring population of human being. But the uneven distribution causes food shortage in some parts of the world when other places have too much nitrogen. Both situations have negative effects on the environment. To effectively solve this problem, the excess nitrogen needs to be regarded as a serious pollution, also the nitrogen fertilizer needs to be used with higher efficiency and nitrogen has to be more evenly distributed around the world to meet the need of food, especially in the area with severe food shortage problems.
© Ling-Hsiang Chen. The author grants permission to copy, distribute and display this work in unaltered form, with attribution to the author, for noncommercial purposes only. All other rights, including commercial rights, are reserved to the author.
[1] J.W. Erisman et al., "Reduced Nitrogen in Ecology and the Environment," Environ. Pollut. 150, 140, (2007).
[2] S. Fields, "Global Nitrogen, Cycling out of Control," Environ. Health Perspect. 112, A556 (2004).
[3] E. B. R. Perotti and A. Pidello, "Plant-Soil-Microorganism Interactions on Nitrogen Cycle: Azospirillum Inoculation," in Advances in Selected Plant Physiology Aspects, ed. by G. Montanaro (InTech, 2014), p. 189.
[4] P. M. Vitousek et al., "Human Alteration of the Global Nitrogen Cycle: Causes and Consequences," Ecological Society of America, Spring 1997.
[5] "Excess Nitrogen in the Environment," in UNEP Year Book 2014, United Nations Environmental Programme, 2014, p. 6.