 |
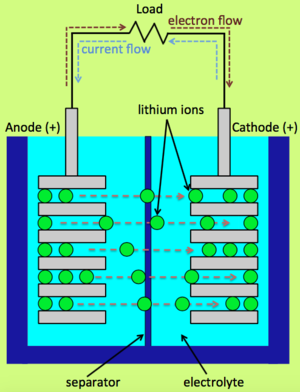
| Fig. 1: As lithium ions travel from the anode to the cathode, electrons provide current to the load. (Source: A. Asundi) |
With ever growing energy demands and a rapid depletion of fossil fuels, it is becoming increasingly important to utilize renewable energy resources. Methods for extracting energy from sustainable sources like the sun and wind are being extensively researched. However, one of the biggest issues that arises when trying to develop ways to harness these energy sources is that they are intermittent. The sun only shines during certain hours of the day and the wind is unpredictable and sporadic. On the other hand, humans demand energy throughout the entire day, and energy demands often peak when renewable resources are not available. The discrepancy between the energy supply and demand motivated the development of batteries, which can be charged and discharged to appropriately meet energy demands. This article will discuss the development of polymers that have demonstrated potential applications as battery electrodes.
A battery consists of three main components: the anode, cathode, and electrolyte. Traditional lithium ion batteries use lithium-metal compounds as the cathode material and a carbon-based anode material. Recently, conducting polymers have been studied as potential cathodes for batteries. Since polymers are lightweight and flexible, they show promise as batteries for a variety of applications and devices.
A charged battery contains cations, typically lithium ions, intercalated into the anode. During discharge, these cations travel from the anode to the cathode through the electrolyte, as shown in Fig. 1. At the same time, electrons travel from the anode to the cathode through an external circuit, thereby providing power to the load. The battery can be recharged by replacing the load with a potential, which drives the cations and electrons in the opposite direction.
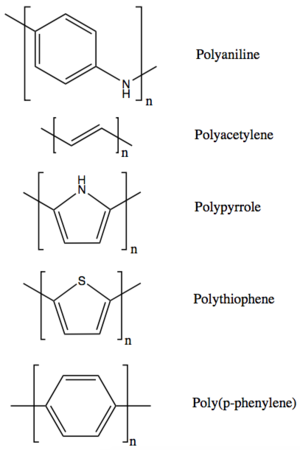 |
| Fig. 2: Chemical structures of some polymers that have been used as battery electrodes. (Source: A. Asundi) |
A polymer is a large molecule made up of a chain of smaller molecules, called monomers. Common examples include rubber, nylon, silicone, starch, and DNA. The properties of a polymer can vary greatly depending on the monomer unit and the way these monomers are attached together. Because of this, polymers have been widely studied for a variety of applications.
Battery electrodes require high conductivity, to allow electrons to charge and discharge easily, and high lithium ion intercalation capacity, to allow for maximum charge storage. Polymers for battery electrodes are made with the appropriate monomers and connectivity to meet these design requirements. Several polymers have shown promise as battery electrodes, including polyaniline, polyacetylene, polypyrrole, polythiophene, and poly(p-phenlyene) (Fig. 2). [1] These polymers are made up of conducting monomers and form hydrogels, highly hydrated polymers with three-dimensional microstructures similar to natural tissues. The combination of conductivity and microstructure allows for facile ion transport. The polymers also have a good interface with the electrolyte due to their hydration properties.
Polymer synthesis typically occurs through three steps: initialization, propagation, and termination. During the first step, an initiator molecule begins the polymerization by reacting with a monomer and forming an unstable, reactive species. The reactive monomer forms a bond with another monomer. This process continues and a long chain of monomers is formed. Once the reaction terminates, the polymer chain no longer grows.
Synthesis of polymer electrodes is very straightforward. For example, at the laboratory scale, polyanilne electrodes were made by simply mixing a solution containing the initiator with a solution containing aniline monomers. [2] The simplicity of electrode synthesis is extremely important for the practical viability of polymer electrodes, as it allows for the process to be easily scaled to industrial levels.
There are many ways to measure the performance of battery electrodes. The ability of an electrode to stored electricity is measured by the capacitance, which is defined as the amount of charge the electrode holds for a given voltage. Capacitance is typically measured through cyclic voltammetry. In this technique, the voltage across the electrode is cycled between two values, with the voltage varying linearly with time. The current response of the electrode is measured. For a capacitor, the current measured equals the capacitance times the rate of change of the voltage. It is therefore possible to calculate the capacitance of an electrode from the current-voltage relation measured in cyclic voltammetry. The kinetics of cation intercalation in the electrode can also be studied using cyclic voltammetry. By varying the rate of change of the voltage, it is possible to probe different intercalation timescales, and thus understand how quickly the electrode can be charged or discharged.
Electrode stability is also extremely important for practical applications. Ideally, it should be possible to charge and discharge the battery many times before needing to replace it. Stability is measured by performing cyclic voltammetry over many cycles and measuring the change in capacitance relative to the first cycle. Such measurements have been done on polyaniline in an acid electrolyte by Pan et al, who found that 83% of the original capacitance was retained after 10,000 cycles. [3]
Polymer batteries with lithium ion electrolytes are used in several applications since they are lightweight and can be made in almost any shape. Though they tend to be more expensive than conventional batteries, polymer batteries have been used in radio controlled cars, mobile phones, and tablet computers, where low weight and flexibility are more important than cost. Lithium polymer batteries are also being tested in electric vehicles.
Research on polymer batteries must continue in many different directions to improve their performance. At the laboratory scale, studies can be done on different electrolyte compositions, rather than lithium-based electrolytes. Self-discharge properties can also be researched to mitigate power loss when the battery is not in use. At the industrial level, polymers synthesized by large scale processes must be studied under realistic operating conditions. Such investigations will expand the viability and potential applications of polymer batteries.
© Arun Asundi. The author grants permission to copy, distribute and display this work in unaltered form, with attribution to the author, for noncommercial purposes only. All other rights, including commercial rights, are reserved to the author.
[1] P. Novak et al, "Electrochemically Active Polymers for Rechargeable Batteries," Chem. Rev. 97, 207 (1997).
[2] J. Stejskal and R. G. Gilbert, "Polyaniline. Preparation of a Conducting Polymer," Pure Appl. Chem. 74, 857 (2002).
[3] L. Pan et al, "Hierarchical Nanostructured Conducting Polymer Hydrogel with High Electrochemical Activity," Proc. Nat. Acad. Sci. (USA) 109, 9287 (2012).