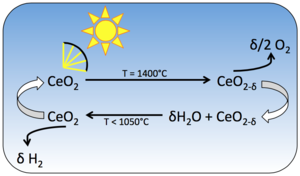
 |
| Fig. 1: Schematic of solar driven thermo-chemical cycling of doped ceria. Water is split and hydrogen fuel is produced during the low temperature step. After Le Gal et al. [7] |
One of the biggest challenges that society will face in the coming decades is developing a sustainable method of producing energy. The sun is a promising source of renewable energy - in 2006 it was estimated that the amount of energy the Earth receives from sunlight in one hour would be enough to fill mankind's energy budget for a full year. [1] As energy demands continue to rise, the sun becomes increasingly attractive as an alternate energy source. The key to harnessing the sun's energy is to convert solar radiation into fuels that can be used more readily. One strategy is to concentrate solar energy in the form of heat to drive the water splitting reaction:
Analyzing the thermodynamics of this process reveals that temperatures above 3000°C are needed to convert more than 50% of the water. [2] Such high temperatures are difficult to achieve and prohibitive for many materials. This motivates the need to develop materials that can facilitate this water splitting reaction at lower temperatures.
One material that has shown promise in thermochemical water splitting is the ceramic material ceria (CeO2). [3] Ceria exhibits a property called non-stoichiometry: at high temperatures (about 1000°C) the material releases oxygen without undergoing a phase change to form the reduced oxide CeO2-δ. The oxygen release is reversible, which means that at low temperatures, the material can take up oxygen to return to the stoichiometric state, CeO2 (Fig. 1). This property of ceria is utilized in the two-step water splitting reaction
Ceria is heated to 1000°C and releases oxygen to form the non-stoichiometric oxide CeO2-δ.
The oxygen deficient material is cooled down in the presence of water vapor. In order to return to the stoichiometric state, CeO2, the material takes the oxygen atom from the water molecule, thus forming hydrogen gas.
After one complete temperature cycle, water has been split into hydrogen and oxygen.
Ultimately we are interested in the amount of hydrogen that can be produced in a given amount of time per gram of ceria. The rate of hydrogen production depends on the thermodynamics and the kinetics of the thermochemical cycle. The thermodynamics dictate the amount of hydrogen that can be produced in a single cycle, while the kinetics govern the amount of time required to complete one cycle. The design of a thermochemical water splitting reactor also plays an important role in dictating the rate of hydrogen production, but discussion of reactor design will be omitted in this article. The thermodynamics and kinetics will be examined in more detail in the next two sections.
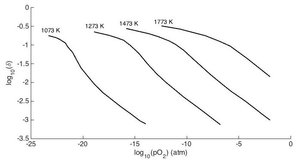 |
| Fig. 2: Dependence of non-stoichiometry on temperature and oxygen partial pressure in ceria. After Panlener et al. [9] |
The thermodynamic parameter that governs the amount of hydrogen produced is the non-stoichiometry, δ. We can see in the chemical reactions above that one cycle produces delta moles of hydrogen. The oxygen non- stoichiometry is in turn a function of two controllable parameters: the temperature of the material and the partial pressure of oxygen in the environment. In particular, as temperature increases, more oxygen atoms escape from the lattice and the non-stoichiometry increases. As the partial pressure of oxygen in the environment increases, it becomes less favorable for oxygen to be in the air and more favorable for it to be in the material, so the non-stoichiometry decreases. If we know how the non-stoichiometry varies as a function of the temperature and oxygen partial pressure, then we know all the thermodynamic information relevant to the thermochemical cycle. In other words, once we specify the temperatures and oxygen partial pressures of the first and second steps of the thermochemical cycle, we will be able to calculate how much hydrogen is produced in one cycle.
The behavior of oxygen non-stoichiometry as a function of temperature and oxygen partial pressure is typically measured through thermogravimetric analysis (TGA). In TGA, the mass of a sample of ceria is tracked under different temperatures and oxygen partial pressures. The change in the sample mass relative to a reference state can be related to the change in the number of oxygen atoms in the ceria lattice. Thus it is possible to calculate the non- stoichiometry behavior from the measured mass changes. Fig. 2 shows the dependence of non-stoichiometry on temperature and oxygen partial pressure. For the first step in the thermochemical cycle, it is important to obtain a large stoichiometry so that a lot of water can be split when the sample returns to the stoichiometric state during the second step. Therefore, the ceria must be cycled between a state of high temperature and low oxygen partial pressure, and a state of low temperature and high water vapor pressure.
The incorporation of gas phase oxygen into the lattice sites of ceria occurs through two steps. First, gaseous oxygen must adsorb onto the surface of ceria. Second, adsorbed oxygen atoms must diffuse through the bulk ceria material to reach a vacant lattice site. This gives rise to two kinetic parameters relevant to thermochemical cycling: the surface reaction constant, ks, and the bulk oxygen diffusivity, D0. During thermochemical cycling the non-stoichiometry changes at a rate determined by these kinetic parameters. Just like non-stoichiometry, ks and D0 are functions of temperature and oxygen partial pressure. It is necessary to understand how the kinetic parameters change during the thermochemical cycle in order to determine the rate of hydrogen production. Higher values of ks and D0 imply shorter thermochemical cycling times and faster hydrogen production.
Measurement of the kinetic parameters is more complicated than non-stoichiometry, but can be achieved with good precision using a technique called electrical conductivity relaxation (ECR). [4] Many physical, chemical, and electrical properties of ceria are functions of non-stoichiometry, which in turn is a function of temperature and oxygen partial pressure. One such property is the electrical conductivity of ceria. If a sample of ceria is maintained at a particular temperature and oxygen partial pressure, its electrical conductivity will equilibrate at a certain value. If the oxygen partial pressure is then changed to a new value the non-stoichiometry will change at a rate governed by the kinetic parameters. This change can be tracked by measuring the behavior of electrical conductivity over time. Theoretical models have been developed to describe the rate of this equilibration process in terms of ks and D0. [5] By fitting these theoretical models to the measured ECR behavior of the material it is possible to extract the values of the kinetic parameters.
Some preliminary economic evaluations of thermochemical cycling using solar energy as the heat source have been performed. [6] These assessments suggest that thermochemical hydrogen production is competitive with water electrolysis and could become competitive with fossil fuels. However, many uncertainties remain regarding the economic viability of solar thermochemical water splitting. In particular, since many components are still in the early stages of development, the investment costs are unknown, and the effects of economies of scale have not been studied. Further examination of the economics is required to fully assess the viability of thermochemical hydrogen production.
Thermochemical water splitting by ceria to produce hydrogen represents a promising source of sustainable energy. In order for this method of fuel production to see practical applications, it must become competitive with currently used fuel sources: coal, oil, and natural gas. Although thermochemical water splitting is not currently competitive with other energy sources, there is room for significant improvements. Doping ceria with other metals like Samarium, Zirconium, Gadolinium, and Lanthanum, alters the thermodynamic and kinetic properties and can improve the rate of hydrogen production. [7] The physical structure of the material can also be tailored towards improved fuel production rates. [8] Further research into thermochemical water splitting materials could potentially give this sustainable fuel production method practical applicability.
© Arun Asundi. The author grants permission to copy, distribute and display this work in unaltered form, with attribution to the author, for noncommercial purposes only. All other rights, including commercial rights, are reserved to the author.
[1] N. S. Lewis and D. G. Nocera, "Powering the planet: Chemical Challenges in Solar Energy Utilization," Proc. Nat. Acad. Sci. (USA) 103, 15729 (2006).
[2] T. Nakamura, "Hydrogen Production from Water Utilizing Solar Heat at High Temperatures," Solar Energy, 19, 467 (1977).
[3] W. C. Chueh and S. M. Haile, "A Thermochemical Study of Ceria: Exploiting an Old Material for New Modes of Energy Conversion and CO2 Mitigation," Phil. Trans. R. Soc. A, 368, 3269 (2010).
[4] C. B. Gopal and S. M. Haile, "An Electrical Conductivity Relaxation Study of Oxygen Transport in Samarium Doped Ceria," J. Mater. Chem. A, 2, 2405 (2014).
[5] M. W. Barsoum, Fundamentals of Ceramics (McGraw-Hill, 1997).
[6] A. Steinfeld, "Solar Hydrogen Production via a Two-Step Water-Splitting Thermochemical Cycle Based on Zn/ZnO Redox Reactions," Int. J. Hydrogen Energy, 27, 611 (2002).
[7] A. Le Gal and S. Abanades, "Dopant Incorporation in Ceria for Enhanced Water-Splitting Activity During Solar Thermochemical Hydrogen Feneration," J. Phys. Chem. C, 116, 13516 (2012).
[8] M. Melchionna and P. Fornasiero, "The Role of Ceria-Based Nanostructured Materials in Energy Applications," Mater. Today, 17, 349 (2014).
[9] R. J. Panlener, R. N. Blumenthal, and J. E. Garnier, "A Thermodynamic Study of Nonstoichiometric Cerium Dioxide," J. Phys. Chem. Solids. 36, 1213 (1975).