
 |
| Fig. 1: A hungry poison waiting for a nuclear reactor to stop! |
Xenon is a noble gas that has an atomic number of 54 and an atomic mass of 131.3 g/mol. It has nine stable isotopes and seven known radioactive isotopes. [1] During a nuclear fission reaction process, a fissile nuclear fuel such as U-235 undergoes a chain of reactions as a result of capturing a thermal neutron. The captured neutron distorts the U-235 nucleus and the electrostatic repulsion force overcomes the nuclear attraction causing a split into two fission fragments. [2] This process is accompanied by energy release in the form of gamma rays as well as neutron emission. Some of the neutrons are released spontaneously while smaller numbers are delayed due to radioactive decay of certain fragments. The fission process produces several neutrons that play major role in sustaining the chain reactions. The fission process releases fragments of different mass numbers mainly around 90 and 140, most of which are radioactive. [3] These fission fragments would decay, each with different half-life, to reach stable end products. Therefore, there would be a series of fragments decay that are inevitable. It is important to keep the chain reaction sustained to ensure neutrons growth to the desired level to deliver the required power output.
One critical stage of the fission reaction is the formation of I-135, a subsequence of Te-135 beta decay. The I-135 is a weak neutron absorber and has a half-life of 6.7 hours. The beta decay of I-135 to Xe-135 introduces a very powerful neutron absorber product. Xe-135 has a large cross section of 2.6 million barns. [4] This is about 5128 times higher than the thermal cross-section of U-235, which is 507 barns. [4] A barn is equivalent to 10-28 m2. This makes it an effective neutron absorber with a high interaction probability with the thermal neutrons. Xe-135 is produced with about 5% probability from every fission process but with 95% probability from I-135 decay. [5] Xe-135 has a half-life of 9.1 hours. [4] Increases in Xe-135 concentration in the reactor stops the neutron growth due its large cross-section. Absorption causes the formation of a stable isotope, Xe-136. [5] The neutron population decreases and the chain reaction stops. The reactor eventually shuts down due to xenon accumulation. Understanding how to deal with the produced Xe-135 is essential to keep the reaction chain sustainable and the neutron flux at desired rate.
When operating a new reactor, the startup stage initially has no trace xenon, and the reactor is maintained critical at a low power level. Small traces of Xe-135 are detected afterwards as a direct product of fission process. As the power level is increased, Xe-135 starts to build up. It eventually reaches an equilibrium level. [5] Negative reactivity is delivered due to Xe-135 growth, and it is proportional to the power level. To maintain criticality, the control rods (absorbers) are withdrawn so the reactor remains critical. This keeps the negative impact of the Xe-135 under control. The destruction of Xe-135 occurs mainly via neutron absorption. Most of it is burned off due to neutron flux forming Xe-136 (weak absorber) and the rest due to beta decay forming Cs-135. [5]
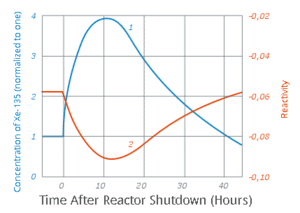 |
| Fig. 2: Xe-135 concentration in the reactor and neutrons reactivity. (Source: Wikimedia Commons) |
The poisoning of Xe-135 arises when the reactor is shut down, see Fig.2. The neutron flux (reactivity) decreases drastically (point 2 in Fig.2) while Xe-135 production increases due to the continuous decay of I-135 see Fig.2. The poisoning increases, therefore, and reaches a peak value few hours after shutting down the reactor, see point 1 in Fig.2. [5] Once the reactor enters what is known as the xenon dead time it would be impossible to restart it. The poison must be allowed to to decay, and such a process may take few days before the restart becomes possible. [5] As a result, a shortage in power supply can suddenly occur to major electrical grids leading to blackout.
There were many bad actors in the famous Chernobyl disaster that occurred in Ukraine in 1986. The incident occurred at Unit 4 reactor of the type RBMK-1000 graphite that had 1000 MWe power output. While there were several flaws in the reactor mechanical design and absence of containment and safety measures, the design of the control system did not account for all possible scenarios. The accident was sparked when the nuclear reactor was shut down for testing at low power, 720 MW. Xe-135 poisoning started to accumulate on the fuel rods and the thermal power kept decreasing to 30 MW. The control rods were withdrawn accordingly to increase neutron reactivity and hence the thermal power. This eventually caused the reactor to become thermo-hydraulically unstable. The complications occurred after that could not be rectified even after reinserting the control rods. The improper handling of the reactor during Xe-135 poisoning by lowering the thermal power at levels insufficient for neutron flux to burn up the Xe-135 was the trigger for the following consequences. This was the role of Xe-135 in Chernobyl disaster. [6]
The thermal power increased to 200 MW after removing the control rods. The number of water pumps used to feed the reactor fell from 8 to 4 during testing that caused steam bubbles (voids) to form in the cooling water that in turn increased the reaction rate rapidly. With a positive void coefficient, one of the design's safety flaws, the reactivity increases as a response to the increase of steam voids. [6] The result was a tremendous increase in thermal power that burned all the Xe-135 and kept increasing to reach 30,000 MW thermal power. [2] Efforts to reinsert the control rods to decrease the power level were useless. The whole reactor eventually exploded. Note that there were some other factors that led to this accident such as the slow rods movement and lack of water fail-safe system.
© Khalid Alnoaimi. The author grants permission to copy, distribute and display this work in unaltered form, with attribution to the author, for noncommercial purposes only. All other rights, including commercial rights, are reserved to the author.
[1] J. Daintith, Ed., A Dictionary of Chemistry, 6th Ed. (Oxford U. Press, 2008).
[2] R. L. Murray, Nuclear Energy, 6th Ed. (Butterworth-Heinemann, 2008), Chapter 6.
[3] D. C. Tayal, Nuclear Physics (Himalaya Publishing House, 2009), p. 581.
[4] W. M., Stacey, Nuclear Reactor Physics, 2nd Ed. (Wiley-VCH, 2007), p. 213.
[5] "Nuclear Physics and Reactor Theory," U.S. Department of Energy, DOE Fundamentals Handbook, DOE-HDBK-1019, Vol. 1 and Vol. 2, January 1993, p. 193.
[6] M. V. Malko, "The Chernobyl Reactor: Design Features and Reasons for Accident," in "Recent Research Activities About the Chernobyl NPP Accident in Belarus, Ukraine and Russia," ed. by T. Imanaka, Research Reactor Institute, Kyoto University, July 2002, p. 11.