 |
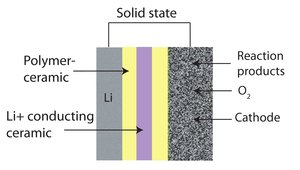
| Fig. 1: Schematic of solid-state type Lithium-air battery design. (Source: Wikimedia Commons) |
Energy consumption is becoming one of the most essential features of nowadays society. Hence energy storage turns out to be especially important. Among the myriad energy-storage technologies, lithium batteries play an increasingly vital role because of their high specific energy densities based on both weight and volume. However, the fully-developed lithium-ion batteries are gradually falling behind of the demands of target market like long-term electrical transportation. [1] Thus, exploring new battery chemistry and materials beyond the horizon of lithium-ion batteries is a urgent but formidably challenging task. By employing redox between lithium and light materials with low electrochemical equivalent such as oxygen, lithium-oxygen/air (Li-O2) batteries are gaining great attention as they can deliver energy density as high as 3500 Wh kg-1 or 3400 Wh L-1. [2] However, there are several problems to be addressed before Li-O2 batteries could lighten the future world.
For the negative electrodes in lithium-air batteries, the common issues are the unstable lithium/electrolyte interphase and hazardous dendrites, which are similar to other metal-anode-involved systems. For the positive electrodes, however, the problems are more specific: (1) sluggish kinetics of oxygen reduction reaction; (2) complex transfer behavior in multiphase (gas/liquid/solid) region; (3) huge charging barrier of converting peroxide back to oxygen; (4) severe side reactions between conductive carbon agents, organic compounds, and highly active superoxide. [3] The former three aspects have been well addressed on some level by rationally designing oxygen electrode structures, including highly efficient electrocatalysts and advanced carbon network. However, the side reactions are still intrinsically inevitable. As a result, the electrolyte and electrodes degrade rapidly. Their decomposed products in turn enlarge the charging barrier. Hence the performances of current lithium-air batteries are irreversible, unstable, and unsustainable.
To address the key issues of stability and sustainability, there are two major concepts: (1) routine cells with liquid electrolyte but inert electrode scaffolds instead of unstable carbon and (2) all-solid-state battery configurations (Figure 1). For the liquid cells, the key component in conventional oxygen electrode of carbon can be replaced by conductive but superoxide resistant materials so that the corresponding side reactions can be alleviated. For example, Peng and Bruce et al. reported a porous gold electrode where Li2O2 can reversibly form/decompose. An unprecedented stability of 95% capacity retention for 100-cycle full charge/discharge was achieved. [4] To further improve the performance and reduce the cost, they developed the system to lighter and cheaper materials like TiC and greatly reduced the side reactions as well. [5] Li et al also prepared a carbon-free oxygen electrode by loading Ru catalysts on indium tin oxide (ITO) support. Such a Ru/ITO electrode exhibited much improved stability and reduced charging barrier. [6] On the other hand, inorganic or polymer electrolytes, comparing to vulnerable organic liquid electrolytes, are expected to be stable and compatible with highly oxidizing per-/superoxides. [7] Thus, they are selected to prove the concept of all-solid-state Li-O2 batteries. For example, Kitaura et al integrated Li-Al-Ti-PO4 (LATP) solid electrolyte particles into carbon nanotube (CNT) network as oxygen electrodes, in which electron/lithium ion was transferred through CNT/LATP networks respectively and oxygen was reduced to form Li2O2 at the interface. [8] Side reaction was greatly eliminated this way.
Liquid or solid? The above two concepts are both effective and efficient. However, some concomitant problems are also derived. Inert electrode scaffolds such as porous gold, TiC, and ITO indeed endow the Li-O2 batteries long-term sustainability. However, the use of heavy element to replace light carbon increases the weight of electrodes, and hence reduces the overall energy density. Solid electrolytes are highly resistant and safe. But their room-temperature ionic conductivities are still 1-2 orders lower than their liquid counterparts. Hence the charging barrier is increased and the energy efficiency is lowered. The two appealing directions are thus producing (1) conductive, light, and chemically stable electrode scaffolds and (2) superionic conductors as superior solid electrolytes. In addition, considering the issues on the anode side, solid Li-O2 battery might be a more promising destination.
© Xinyan Liu. The author grants permission to copy, distribute and display this work in unaltered form, with attribution to the author, for noncommercial purposes only. All other rights, including commercial rights, are reserved to the author.
[1] M. Armand and J.-M. Tarascon, "Building Better Batteries," Nature 451, 652 (2008).
[2] P. G. Bruce et al., "Li-O2 and Li-S Batteries With High Energy Storage," Nat. Mater. 11, 19 (2012).
[3] L. Johnson et al., "The Role of LiO2 Solubility in O2 Reduction in Aprotic Solvents and its Consequences For Li-O2 Batteries," Nat. Chem. 6, 1091 (2014).
[4] Z. Peng et al., "A Reversible and Higher-Rate Li-O2 Battery," Science 337, 563 (2012).
[5] M. M. O. Thotiyl et al., "A Stable Cathode for the Aprotic Li-O2 Battery," Nat. Mat. 12, 1050 (2013).
[6] F. Li et al., "Ru/ITO: A Carbon-Free Cathode for Nonaqueous Li-O2 Battery," Nano Lett. 13, 4702 (2013).
[7] F. Li, H. Kitaura, and H. Zhou, "The Pursuit of Rechargeable Solid-State Li-Air Batteries," Energy Environ. Sci. 6, 2302 (2013).
[8] H. Kitaura ad H. Zhou, "Electrochemical Performance of Solid-State Lithium-Air Batteries Using Carbon Nanotube Catalyst in the Air Electrode," Adv. Energy Mat. 2, 889 (2012).