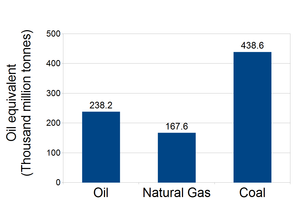
Coal is a fossil fuel and one of the most important energy resources of modern society. In the United States, coal is the largest source for electricity generation, consistently producing more than 30 percent of the country's electricity. [1] The total world coal reserve also contains a tremendous amount of energy, much larger than that of oil or natural gas. [2] Fig. 1 shows that the world coal reserve is equivalent to almost twice the world oil reserve. [2] The reserve-to-production (R/P) ratio is the length of time calculated by dividing the remaining reserve of a resource by its current production rate. [2] The R/P ratios of the world's total oil and natural gas in 2013 were only 53.3 years and 55.1 years respectively, whereas the R/P of the world's coal in 2013 was 113 years. [2] Therefore I think the numbers indicate that coal will likely last much longer than oil and natural gas, thus having a profound influence on the future of energy. Since the longevity of coal is important for the future, one natural question is: how can we extract all the coal there is? Like all other fossil fuels, coal is buried deeply underground and much of the reserve is hard to access. Underground Coal Gasification (UCG) provides a means to extract reserves otherwise unrecoverable. [3] UCG can potentially increase the recoverable reserves in the U.S. by 300- 400%. [3] This technology also has other merits such as eliminating mining accidents. [4] There are, however, also major environmental concerns about UCG. [4] This report intends to introduce the process of UCG, its advantages and drawbacks.
Underground Coal Gasification is a chemical process that converts underground coal into a mixture of gaseous products in-situ (meaning the process happens in the coal reservoirs). [4] The main products include methane, hydrogen, carbon monoxide and carbon dioxide. [5] Methane is a natural gas and a combustible energy source. A mixture of hydrogen and carbon monoxide is called a syngas and can be used to produce synthetic natural gas or liquid fuel. [6] In UCG, wells are drilled down into the coal seam to inject and extract gas. [4] By pumping air or oxygen into the coal seam, coal can essentially be burned underground under high pressure conditions to produce the gases mentioned above, which are then extracted through separate wells to the surface. [5]
UCG has many advantages over conventional mining. The most obvious advantages are: UCG eliminates conventiaonl mining and all the dangers associated with it, such as mine collapse and asphyxiation; [4] UCG converts conventionally unrecoverable coal into useful resources such as syngas. [3] Surface coal gasifiers are no longer needed, thus reducing the cost of infrastructure. [4] The capital and environmental costs of coal transportation are also eliminated with UCG. [4] Last but not least, UCG has the potential to be more environmentally friendly than conventional coal production and surface gasification because the pollutants are easier to control. [4] The ash stays underground and the process of UCG produces less greenhouse gases than the combined operation of coal mining and surface combustion. [4] The carbon dioxide produced in UCG can also potentionally be stored underground, thus reducing emission to the atmosphere. [4]
There are two major concerns about UCG: groundwater contamination and ground subsidence. [3] Groundwater can be contaminated by gaseous or aqueous contaminants through several "leaching" mechanisms under the high pressure inside the UCG reactor. [5] These mechanisms and the chance for contamination are very dependent on the geologic and hydrologic features of the site. [5] Therefore careful selection of the cavity left behind from transferring matter to the surface during the UCG process. [3] A series of UCG trials conducted by the Lawrence Livermore National Laboratory at Hoe Creek resulted in significant subsidence and groundwater contamination. [5] On the other hand, the Chinchilla project in Australia reported neither groundwater contamination nor subsidence after burning more than 20000 tonnes of coal. [5] Since the trial data is still very limited, the risks of these hazards require further study. Under the strict site selection criteria posed by environmental concerns, however, the number of sites suitable for UCG may be greatly reduced.
UCG has both promising applications and significant concerns. Even though the idea of UCG was suggested as early as the 1800s, the economics and consequences of large scale UCG are still relatively uncertain. Nevertheless the vast potential benefits of UCG should be seriously considered in the age of the energy crisis. [4] In particular, the U.S. has roughly 27 percent of the total world's coal reserve. [2] Therefore it should be a priority for the U.S. government to research and develop feasible UCG facilities. The potential negative environmental impact of UCG may be avoided through sensible site selection and process control, which further necessitates government and public support for more comprehensive research and testing regarding UCG. [3]
© Zheng Cui. The author grants permission to copy, distribute and display this work in unaltered form, with attribution to the author, for noncommercial purposes only. All other rights, including commercial rights, are reserved to the author.
[1] "Electric Power Monthly with Data for September 2014," U.S. Energy Information Administration, November 2014.
[2] "BP Statistical Review of World Energy," British Petroleum, June 2014.
[3] K. Walter, "Fire in the Hole," Science and Technology Review, Lawrence Livermore National Laboratory, UCRL-TR-52000-07-04, April 2007.
[4] E. Shafirovich and A. Varma, "Underground Coal Gasification: A Brief Review of Current Status," Ind. Eng. Chem. Res. 48, 7865 (2009).
[5] M. Sury et al., "Review of Environmental Issues of Underground Coal Gasification," U.K. Department of Trade and Industry, "DTI/PUB-URN-04/1880, November 2004.
[6] M. R. Beychok, "Process and Environmental Technology for Producing SNG and Liquid Fuels," U.S. Enironment Protection Agency EPA-660/2-75-011, May 1975.