 |
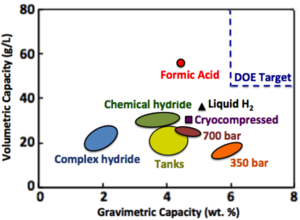
| Fig. 1: Energy densities of various kinds of hydrogen storage. (Source: Wikimedia Commons) |
As the world tries to move away from fossil fuels, hydrogen is thought to be an alternative energy carrier because it can be efficiently used to generate electricity in fuel cells without pollution. [1,2] To implement this fuel cell based hydrogen economy, however, we need to have practical solutions to controlled storage and release of hydrogen. [2,3] Currently established hydrogen storage technologies include compressing hydrogen at high pressures (350 or 700 bar), liquefying hydrogen at low temperatures (below 20.3 K), or storing hydrogen as chemical/metal hydrides. These technologies, however, suffer from high costs, low energy densities, loss of hydrogen, and safety issues. [4,5]
Recently, scientists in Europe have identified formic acid as one of the most promising materials for hydrogen storage because formic acid is a liquid that contains 4.4 wt.% of hydrogen with a volumetric capacity of 53.4 g/l at standard temperature and pressure. [6,7] Even though its hydrogen content falls short of the milestones (5.5 wt.%) set by the US Department of Energy for 2010, its volumetric capacity surpasses that of most other hydrogen storage materials used today (see Fig. 1). [6] In addition, diluted formic acid (85%) is an easy-to-handle liquid that is relatively non-toxic, non-corrosive, and non-flammable, which is an important consideration for mobile applications.
Hydrogen stored in formic acid (HCOOH) can be released on demand by decomposing formic acid into hydrogen (H2) and carbon dioxide (CO2) on a catalytic surface. [8] The co-product of this process, CO2, can be used as a hydrogen vector by hydrogenating it back to formic acid on a catalytic surface, ultimately establishing a carbon-neutral cycle on earth (see Fig. 2). The latter process, i.e., CO2 hydrogenation, has long been studied and many efficient procedures have been already developed. [9,10] Accordingly, most research into developing formic acid as a hydrogen storage material is focused on finding the catalyst materials for formic acid decomposition.
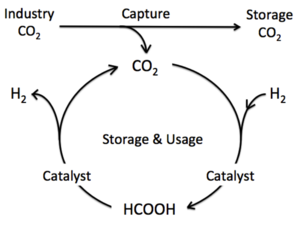 |
| Fig. 2: Hydrogen storage cycle established by using formic acid as a hydrogen storage material. (After Boddien and Junge. [19]) |
To be used as a hydrogen storage material, formic acid needs to be decomposed via dehydrogenation (HCOOH → H2 + CO2 ) rather than via dehydration (HCOOH → H2O + CO) such that virtually CO-free hydrogen is produced. This is particularly important for direct applications in fuel cells because even a trace amount of CO (100 ppm) in hydrogen feedstock can severely deactivate the Pt-based catalysts that are usually used as the anodes of fuel cells. [11] Accordingly, we need to find catalyst materials that are not only active for formic acid decomposition, but also extremely hydrogen selective (selectively catalyzing the dehydrogenation reaction). In this article, recent achievements in the development of catalysts for formic acid decomposition will be discussed, particularly focusing on the heterogeneous systems.
Recent studies on screening of the materials have shown that the Pt-group metals are highly active for formic acid decomposition. [12] For example, Solymosi and co-workers investigated formic acid decomposition over a range of monometallic nanoparticles (Ir, Pd, Pt, Ru or Rh) supported on carbon at 423 K. [13] All their catalysts showed relatively promising activity, but hydrogen was produced with considerable amounts of CO (much higher than 100 ppm of CO) on all catalysts. (See Table 1.)
|
||||||||||||||||||||||||||||||||||
| Table 1: Experimentally observed catalytic properties of nanoparticles for formic acid decomposition. [13] | ||||||||||||||||||||||||||||||||||
Adding a secondary metal to an active phase of a catalyst (in this case, the Pt-group metal) is a well-known means to alter catalytic properties because it modifies the electronic structure of the active phase by alloy formation. Many researchers have taken this approach to improve hydrogen selectivities of the Pt-group metals. The improvements in hydrogen selectivity were particularly significant for Pd-Au and Pd-Ag alloy catalysts which produced hydrogen with CO coproduction well below 100 ppm under ambient conditions. [14-17]
Another research worthy of attention was done by Ojeda et al., which showed that highly dispersed gold nanoparticles supported on Al2O3 can exhibit considerable formic acid decomposition activity, producing virtually CO-free hydrogen. [18] The intrinsic catalytic activity of the gold nanoparticles began to exceed that of the Pt nanoparticles decreasing particle size. This was also confirmed when they observed that the active metal sites are not situated on TEM-visible gold particles but rather on much smaller gold domains (e.g. isolated gold atoms).
Considering all the achievements made in catalyst development, using formic acid as a hydrogen storage material seems to be a viable technology that offers many benefits as compared to the conventional hydrogen storage technologies. However, it is important to note that the catalyst materials developed so far are heavily dependent upon expensive elements. To commercialize the technology, we are still in need of novel catalyst materials that are not only active and selective for formic acid dehydrogenation but also potentially inexpensive.
© Jong Suk Yoo. The author grants permission to copy, distribute and display this work in unaltered form, with attribution to the author, for noncommercial purposes only. All other rights, including commercial rights, are reserved to the author.
[1] J. A. Turner et al., "Sustainable Hydrogen Production," Science 305, 972 (2004).
[2] L. Schlapbach and A. Züttel, "Hydrogen-Storage Materials For Mobile Applications," Nature 414, 353 (2001).
[3] R. M. Navarro, M. A. Peña and J. L. G. Fierro, "Hydrogen Production Reactions From Carbon Feedstocks: Fossil Fuels and Biomass," Chem. Rev.107, 3952 (2007).
[4] M. Felderhoff et al., "Hydrogen storage: The Remaining Scientific and Technological Challenges," Phys. Chem. Chem. Phys. 9, 2643 (2007).
[5] S. Enthaler, J. von Landermann and T. Schmidt, "Carbon Dioxide and Formic Acid - The Couple For environmental-Friendly Hydrogen Storage?," Energy Environ. Sci. 3, 1207 (2010).
[6] M. Grasemann and G. Laurenczy, "Formic Acid as a Hydrogen Source - Recent Developments and Future Trends," Energy Environ. Sci. 5, 8171 (2012).
[7] A. Boddien et al., "CO2-Neutral Hydrogen Storage Based on Bicarbonates and Formates," Angew. Chem. Int. Ed. 50, 6411 (2011).
[8] Q.-Y. Bi et al., "Efficient Subnanometric Gold-Catalyzed Hydrogen Generation via Formic Acid Decomposition under Ambient Conditions," J. Am. Chem. Soc. 134, 8926 (2012).
[9] G. Peng et al., "CO2 Hydrogenation to Formic Acid on Ni(111)," J. Phys. Chem. C 116, 3001 (2012).
[10] D. Preti et al., "Carbon Dioxide Hydrogenation to Formic Acid by Using a Heterogeneous Gold Catalyst," Angew. Chem. Int. Ed. 50, 12551 (2011).
[11] J. S. Yoo et al., "Preparation of a CO-Tolerant PtRuxSny/C Electrocatalyst With an Optimal Ru/Sn Ratio by Selective Sn-deposition on the Surfaces of Pt and Ru," Int. J. Hydrogen Energy 36, 1930 (2011).
[12] D. A. Bulushev, S. Beloshapkin, and J. R. H. Ross, "Hydrogen From Formic Acid Decomposition Over Pd and Au Catalysts," Catal. Today 154, 7 (2010).
[13] F. Solymosi et al., "Production of CO-free H2 From Formic Acid. A Comparative Study of the Catalytic Behavior of Pt Metals on a Carbon Support," J. Catal. 279, 213 (2011).
[14] Y. Huang et al., "Novel PdAu@Au/C Core-Shell Catalyst: Superior Activity and Selectivity in Formic Acid Decomposition for Hydrogen Generation," Chem. Mater. 22, 5122 (2010).
[15] X. Zhou et al., "High-Quality Hydrogen From the Catalyzed Decomposition of Formic Acid by Pd-Au/C and Pd-Ag/C," Chem. Commun., Issue 30, 3540 (2008).
[16] K. Tedsree et al., "Hydrogen Production From Formic Acid Decomposition at Room Temperature Using a Ag-Pd Core-Shell Nanocatalyst," Nat. Nanotechnol. 6, 302 (2011).
[17] S. Zhang et al., "Monodisperse AgPd Alloy Nanoparticles and Their Superior Catalysis for the Dehydrogenation of Formic Acid," Angew. Chem. Int. Ed. 52, 3681 (2013).
[18] M. Ojeda and E.Iglesia, "Formic Acid Dehydrogenation on Au-Based Catalysts at Near-Ambient Temperatures," Angew. Chem. Int. Ed. 121, 4894 (2009).
[19] A. Boddien and H. Junge, "Acidic Ideas For Hydrogen Storage," Nat. Nanotech. 6, 265 (2011).