
 |
| Fig. 1: Photographic image of open pit mine in Australia. (Courtesy of the WISE Uranium Project.) |
The growing concerns over the long-term sustainability of a fossil fuel based energy and increased awareness about global warming have caused a push for alternative energy. While many different energy technologies are competing for the spotlight, nuclear energy may need to be accepted as a valid alternative to fossil fuels. To accommodate the growing need for electrical energy, nuclear electricity generation is expected to grow at more than 1% annually. [1] While efforts are underway to improve the lifetime of these plants and improve uranium reprocessing technology, uranium mining will be important for nuclear electricity to remain a viable long-term option.
The need to find significant uranium deposits for mining has resulted in the development of a variety of survey techniques. However, the most common exploration techniques used for regional prospecting are aerial photography, aeroradiometry, the hydrochemistry of ground water, and gamma-ray surveys for identifying specific drilling sites. [2] While most of the world's uranium is mined in Canada, Australia, and Kazakhstan, the most significant portion of the world's uranium resources are found in Australia. [1] Most uranium is found in the upper crust, occurring naturally as U4+ and U6+ with an average abundance of 2 ppm. [3] Its most naturally occurring mineral is Uraninite (UO2), which typically reacts with oxygen to form U3O8, the mineral pitchblende. Uranium is not usually alloyed into other crystalline compounds due to this high charge state and its extremely large atomic radius, but its oxide is sometimes found within other crystalline compounds such as zircon, thorite, and fluorite, among many other accessory minerals. [3] Because of this, uranium extraction is used to isolate and purify the uranium containing compounds from the raw ore. This paper will focus on the chemistry of uranium leaching techniques, with a brief discussion of mining methods such as open pit mining and underground mining.
Underground and Open pit mines were the most numerous uranium mines in the United States for many years, especially at the beginning of the push for nuclear energy. Fig. 1 shows an image of an open pit mine in Australia. Underground mines were far more prevalent, but both techniques were used because they were techniques developed to mine other materials. Uranium was originally found as a bi- product of Radium and Vanadium mining. As the need for uranium increased, uranium specific mines were opened. However, these mines became more and more expensive as shallow, high grade deposits became depleted and the cost of mining equipment, transportation, and regulations increased. For underground mines, once the site has been identified, a mine shaft is typically drilled down to the ore bed. Since drilling is used in both of these processes, large amounts of water are employed to prevent airborne radiation exposure to laborers. These processes are very similar to bulk mining techniques employed for other materials. The raw material, which is U3O8, must also be analyzed with a beta-gamma detecting instruments to ensure proper levels of uranium content. [2,4] Most of the chemistry of the uranium mining process occurs after the ore has been isolated. These techniques to extract uranium from the ore are referred to as leaching and typically occur at uranium mills.
 |
| Fig. 2: Process scheme for uranium mining and processing to produce useable uranium product, including chemical equations for acid and alkaline leaching process. |
Uranium leaching is the process by which the uranium is extracted from the raw ore by reacting the material with acid or base. Fig. 2 shows a general process flow for the uranium extraction process. Prior to the leaching process, the ore is often given preliminary treatments that can include roasting and grinding. The roasting process is meant to burn carbonaceous species trapped within the ore and make the ore more reactive to the leaching treatments. In some cases, the ore is roasted with salts in order to improve solubility of contaminant metals, such as vanadium, in the leach solution. [4] While, the exact details of the pretreatment process depend on the conditions of the specific uranium mine, all mills some forms of pretreatment prior to leaching. The leaching process is then performed using either an alkaline or acid leaching agent, the selection of which is usually determined by the economics of the process, which depends on the nature of the ore.
Acid leaching has the advantage of being more effective with difficult ores, requiring lower temperatures and leaching times compared to alkaline solutions. It also requires less pretreatment than alkaline leaching, most notably because the particle size from the grinding process does not need to be as small. [4] Acid leaching is sometimes also referred to as heap leaching because the leaching process can be performed on large "heaps" of uranium ore that have been collected from mines. The chemistry of the leaching process revolves around oxidation of the uranium compounds, which is typically achieved using manganese dioxide (MnO2), sodium chlorate (NaClO3), and Fe(II) salts. [4] Fig. 2 shows a typical reaction pathway for the acid leaching process using sulfuric acid. Sulfuric acid is typically used due to the solubility of uranyl sulfate complexes. [4] The reaction is typically performed at slightly elevated temperatures (~60C) and can often release H2, H2S, and CO2 gases during the process. [4] The uranium, which typically begins in the tetravalent state, goes through a series of reactions, eventually leading to the formation of the desired complex, [UO2(SO4)3]4-. While the solubility of this complex makes sulfuric acid a desirable leaching agent, nitric and hydrochloric acid can also be used, but are typically not due to their higher cost and corrosiveness. [4]
While both acidic and alkaline leaching agents are used, alkaline leaching has some significant advantages. Alkaline solutions tend to be more selective to uranium minerals, which means the solution will contain fewer impurities. Consequently, the uranium oxide (commonly called "yellow cake") can be directly precipitated without purification. Furthermore, the solutions are less corrosive and can be recycled without the annoyance of increasing impurity concentrations. Fig. 2 shows a list of reactions involved in the alkaline uranium leaching process. [4] The alkaline leaching process relies on the formation of highly soluble uranyl tricarbonate, UO2(CO3)34-. As in the case of acid leaching, oxidizers are used to maintain the presence of the hexavalent U6+ cation. This can be achieved by simply introducing oxygen into the solution by bubbling air into the solution. [4] The leaching agents used are sodium bicarbonate and sodium carbonate. This prevents formation of uranyl hydroxide compounds. Due to the slower reactivity of the alkaline solutions, increased pressures and temperatures are sometimes used to accelerate the process. [4]
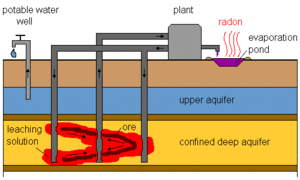 |
| Fig. 3:of in-situ uranium leaching plant. (Courtesy of the WISE Uranium Project.) |
In-situ uranium leaching has been explored as a means of accelerating the uranium production process. The chemistry of these processes is very similar to the extraction methods, except that the leaching solution is pumped into the uranium ore while it is still in the ground. [2,5] Fig. 3 shows a schematic of the in-situ leaching process, in which the leaching solution is pumped into the ore zone containing the uranium ore. The leaching solution is pumped back up into a plant for further processing. A significant concern with in-situ leaching is the contamination of the soil and ground water in the vicinity, as well as the need to process and contain the massive amount of leaching solution. Various treatment methods are used in the vicinity of plant for aquifer restoration, including removal of heavy metals. [2]
Uranium recovery from seawater has been researched as an alternative means of uranium production. Reports indicate that the uranium concentration in seawater is uniformly 3.3 ppb, which is multiple orders of magnitude below even meager ore deposits. [5] It exists in the form similar to that used in alkaline leaching, as a pH dependent carbonate salt. The main impediments to extracting uranium from seawater is the extremely large amount of water that must be processed. Uranium adsorbers and ion-exchange resins have been developed to help remove the uranium, but a significant amount of energy is consumed to pump water through these systems and many studies question the economic feasibility of this method of uranium extraction. [5]
Uranium extraction from ore involves the use of acid or base to break own and extract uranium from the raw ore. These processes require significant amounts of acids or bases, the most commonly used being sulfuric acid and sodium carbonate, respectively. Depending on the leaching agents used, the uranium production process also involves several precipitation and purification steps post leaching that were not covered in this work. The purpose of these steps is to isolate the uranium complexes from other metals and can be done using standard chemical purification processes, such as ion-exchange resins. [4] Alternative methods for uranium production are in development, including in-situ leaching to improve uranium production. As the world's energy needs continue to rise and the nuclear power option becomes increasingly desirable, continued advances in the uranium mining and production may be achieved.
© Ben Weil. The author grants permission to copy, distribute and display this work in unaltered form, with attribution to the author, for noncommercial purposes only. All other rights, including commercial rights, are reserved to the author.
[1] Uranium 2009: Resources, Production and Demand (OECD Publications, 2010).
[2] Uranium Resource/Technology Seminar II (Colorado School of Mines, 1979).
[3] R. A. Rich, U. Petersen and H. D. Holland, Hydrothermal Uranium Deposits (Elsevier, 1980).
[4] Y.-S. Kim, ed., Uranium Mining Technology: Proceedings of a First Conference (U. of Nevada, 1977).
[5] Uranium Extraction Technology: Current Practice and New Developments in Ore Processing (OECD Publishing, 1983).