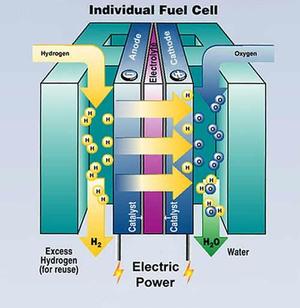
 |
| Fig. 1: The scheme of hydrogen fuel cell. (Courtesy of the U.S. Department of Energy.) |
As the world consumes more and more fossil fuels and the global climate becomes warmer and warmer, people start to reconsider the ways we burn the earth. Renewable energies are like rising stars these days just because we realize that this may be the only way out of the energy and environment crisis. Solar cells, wind energy, global thermal energy, and bio mass, all of them claim that they are the future of the world. Hydrogen, which is the most friendly fuel to the environment and has the largest energy density, came up to the stage because of not only its broad applications, but also the ease to produce. In this paper, I briefly introduce the application of hydrogen as an energy carrier, and explain the catalysts for hydrogen evolution reaction (HER) in detail.
Hydrogen has the largest energy density over any other fuel in the world. When we burn hydrogen in air, the reaction should be:
Which means that 1mol of hydrogen can produce 286kJ energy through reaction with oxygen. Obviously, the energy density of hydrogen is E = 286 kJ/mol × 0.5 mol/g = 143kJ/g, which is much higher than that of many other fuels such as methane. Because of the high energy density, in order to reduce the weight of space shuttle, people choose to use liquid hydrogen as the fuel.
Also another important application of hydrogen as an energy carrier is fuel cell. Hydrogen acts as the anode and splits into protons and electrons under the assistance of catalyst. Protons move from anode towards cathode through a conductive electrolyte, and electrons go through external circuit and meet with protons and oxygen in cathode. Then the product, water, is produced, as well as electric power. The voltage of this cell can be calculated by the Gibbs free energy, ΔG = -237.1 kJ/mol, which gives the voltage applying the Nernst equation E = -ΔG/nF = 237.1kJ/mol2 × 96485C/mol = 1.229 V. [1] The fuel cells contain a large amount of energy, and the product is water, which is totally friendly to the environment. The investigations into the fuel cells are going to make an avenue to the applications of hydrogen as an energy carrier, such as the hydrogen cars and hydrogen power station.
Nowadays there are two ways for us to produce hydrogen. In industry, hydrogen is produced by steam reforming of natural gas (CH4), which is shown as follows:
Most of the hydrogen we use today comes out from this reaction in factories. However, it is familiar to most people that we can also split water to produce hydrogen. As I mentioned in the fuel cell, the output voltage is 1.23 V. This voltage is reversible, which means that 1.23 V is the lowest limit voltage we need to split water into hydrogen and oxygen. However, the voltage is calculated thermodynamically, and kinetically, higher voltage is needed depending on the electrode we use. This topic relates to the catalyst for HER, and in order to explain it clearly, I introduce the Tafel behavior in g electrochemistry briefly.
For HER with acid electrolyte, hydrogen evolves from the reaction 2H + aq + 2e → H2(g). The rate of this reaction is tested by the current through the circuit. The relationship between the current and the voltage we apply is called current-overpotential equation: [1]
in which C0 and CR represent the concentrations of the participants, i0 is the exchange current, α stands for the asymmetry coefficient, f=F/RT, and η represents the overpotential. When the overpotential goes to large negative, the left term in the bracket is much larger than the right one, and the equation can be rewritten as i=i0 e-αnfη. The current increases exponentially with overpotential, which is named as Tafel behavior. From this behavior, we can extract two important parameters, exchange current i0 and Tafel slope 1/αnf by fitting the linear relationship between log(i) and η. These two parameters are the criterions for the catalytic activity of catalysts. In order to be the best catalyst for HER, it should achieve the largest exchange current and the smallest Tafel slope. And until now as we know, platinum is the unbeatable catalyst for HER which has the exchange current density of 4.5 × 10-4 A/cm2, and the Tafel slope as small as 30 mV/decade. [2]
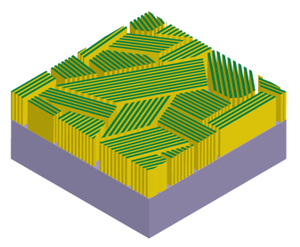 |
| Fig. 2: The Idealized catalyst structure with molybdenum chalcogenide layers aligned perpendicular to the substrate form a fully active edge-terminated film. |
However, as we know, platinum is very scarce in the earth and extremely expensive ($50/g). Scientists have been trying to explore other cheap and abundant materials which are also very active catalysts for HER to substitute platinum. Professor Jens Norskov at Stanford University explored the mechanism of HER by applying the density functional theory calculations. In his work, the free energies of different materials for HER are calculated and MoS2 is comparable with Pt. [3]
The whole process of HER can be divided into two steps: absorption and desorption. First, the protons from the solution are attached to the catalytic sites of the electrode. Then the electrons from the electrode combine with the protons to form hydrogen atoms. Two hydrogen atoms combine together to become a hydrogen molecule. Desorption then takes place and molecular hydrogen leaves from the site of the electrode. One of the two steps may be the rate determine step, which is obvious from the figure above. For Pt, the rate determining step is the desorption step because the Gibbs free energy is uphill. For MoS2, rate determining step changes to the absorption step. We can also find that when the free energy approaches to zero, the catalyst becomes very active because the rate determining step is also very fast.
Searching from the figure above, we find that MoS2 is a good catalyst candidate for HER, which may be comparable with Pt. However, as with bulk MoS2, it is not active for HER at all. The reason can be explained after I show the atomic structure of MoS2 below. [4]
MoS2 has a hexagonal structure consisting of three covalently bonded atomic sheets, which are S-Mo-S in sequence. The planar, two-dimensional layers are linked by van der Waals interaction. Two general types of surface sites are present on this crystal: terrace sites on the basal planes and edge sites on the side surfaces. However after theoretical calculation, the edge sites are found to be active for HER, but not the terrace sites. That is the reason why bulk MoS2 is not an active catalyst for HER because not enough edge sites are exposed. In 2007, Ib Chorkendroff experimentally proved that the edge sites of MoS2 are indeed the active sites for HER, instead of terrace sites. [5] They prepared single layered MoS2 samples on Au(111) substrate, and were characterized by STM. They showed us that the exchange current density, which should be proportional to the density of active catalytic sites, does not have a linear relationship with MoS2 area coverage, but does with MoS2 edge length. This is a very strong argument that it is the edge site that is active for HER.
Depending on the studies about MoS2 above, many scientists began to explore effective methods to expose the edge sites of MoS2 as many as possible, such as MoS2 nanoparticles, nanowires, amorphous MoS2 and so on. [5-8] And if we want to achieve the largest edge site density on a 2-D substrate, vertically standing layers of MoS2 are needed, as shown in Fig. 2. This catalyst design not only offers maximum density of edge sites, but also guarantees efficient charge flow from the conductive support to active surface sites due to the high intra-layer conductivity.
In conclusion, I introduce the application of hydrogen as an energy carrier, and then discuss in detail about the catalysts for hydrogen evolution reaction. Hopefully the hydrogen energy is able to solve both the energy and environment crisis in the world.
© Haotian Wang. The author grants permission to copy, distribute and display this work in unaltered form, with attribution to the author, for noncommercial purposes only. All other rights, including commercial rights, are reserved to the author.
[1] A. J. Bard and L. R. Faulkner, Electrochemical Methods (Wiley, 2000).
[2] B. E. Conway and B. V. Tilak, "Interfacial Processes Involving Electrocatalytic Evolution and Oxidation of H2 and the Role of Chemisorbed H," Electrochim. Acta 47, 3571 (2002).
[3] B. Hinnemann et al., "Biomimetic Hydrogen Evolution: MoS2 Nanoparticles as Catalyst for Hydrogen Evolution," J. Am. Chem. Soc. 127, 5308 (2005).
[4] C. Kisielowski et al., "Imaging MoS2 Nanocatalysts With Single-Atom Sensitivity," Angew. Chem. Int. Ed. 49, 2708 (2010).
[5] T. F. Jaramillo et al., "Identification of Active Edge Sites for Electrochemical H2 Evolution From MoS2 Nanocatalysts," Science 317, 100 (2007).
[6] Y. Li et al., "MoS2 Nanoparticles Grown on Graphene: An Advanced Catalyst for the Hydrogen Evolution Reaction," J. Am. Chem. Soc. 133, 7296 (2011).
[7] Z. Chen et al., "Core-Shell MoO3 Nanowires for Hydrogen Evolution: A Functional Design for Electrocatalytic Materials," Nano Lett. 11, 4168 (2011).
[8] J. D. Benck et al., "Amorphous Molybdenum Sulfide Catalysts for Electrochemical Hydrogen Production: Insights Into the Origin of Their Catalytic Activity," ACS Catal. 2, 1916 (2012).