 |
| Fig. 1: This is the caption. |
At some point in the future we are going to stop burning fossil fuels. This will either be because extraction is too expensive or the environmental cost is too high.
This, then, raises the question of what will take their place. As a means of storing energy, fossil fuels have several key advantages over their main competitors. The most important of these advantages are:
Energy density - both in terms of mass and volume;
Existing infrastructure.
Indeed there are some applications, e.g. flight, for which it is hard to conceive of any alternative power source.
Given the advantages of carbon-based liquid fuels, it may make sense to manufacture them synthetically rather than substitute an inferior energy source. Whether or not we do so will come down to a trade-off between cost and convenience, e.g. battery powered cars may be cheaper to "refuel" but they are unlikely to be as cheap to build or have the range of one powered by gasoline.
The steps in creating green synthetic fuels can be broken down as follows:
Electrolysis of water to create hydrogen;
Capture of carbon dioxide from the atmosphere;
Reduction of carbon dioxide to carbon monoxide;
Conversion of carbon monoxide and hydrogen into liquid fuels (Fischer-Tropsch process).
None of the processes outlined above are novel. Electrolysis of water dates from 1800 and the Fischer-Tropsch process was extensively used in Germany during World War II. [1,2] Some companies are already producing fuels using steps 1 - 4, albeit on a very small scale, just 5 liters over a period of three months. [3]
If the energy used to drive the reactions above comes from a renewable source, then fuels created using this method are entirely carbon neutral - the amount of CO2 released during combustion is equal to the amount captured during production.
It is also possible to create synthetic fuels using just step 4 but with hydrocarbons (e.g. natural gas, coal etc.) as the feedstock. This is generally done when the cost per joule of one form of fossil fuel is very different to the cost per joule of another. A modern example is Qatar. Qatar has some 12% of the worlds proven gas reserves yet no easy way of transporting it to overseas markets. [4] As a consequence Shell have invested some $19bn in a gas to liquid plant, which, as long oil remains above $70 a barrel, will be profitable. [5] Unfortunately this hydrocarbon conversion is very environmentally unfriendly as there is no carbon capture and CO2 is released during the conversion stage.
Given the obvious environmental benefits of using renewable electricity to create all of our liquid fuels synthetically from water and air - why aren't we doing so to a greater extent?
The answer, as ever, is cost. Many of the steps involved in the production of green synthetic fuels are very expensive both in terms of infrastructure and energy requirements. By comparison mining hydrocarbons from the ground, or converting them from one form to another, is very cheap.
Viewed over the long term, the cost of fossil fuels is likely to rise and the cost of renewable energy will fall. So in time there may be a point at which it becomes profitable to simply manufacture our own gasoline rather than extracting it from the ground.
What, though, is this point likely to be? We are interested in the long term efficiency of any conversion process, so simply using current costs is likely to give a very misleading result.
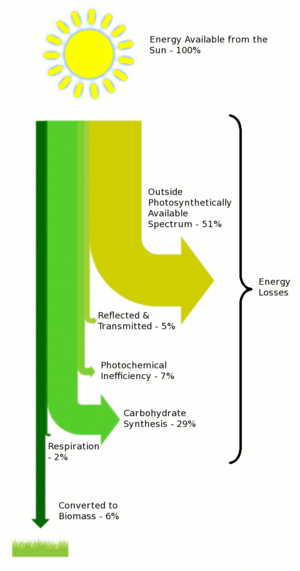
One potential insight comes from plants; they have been converting water and carbon dioxide into biomass for millions of years and the chemical structure of biomass is very closely related to that of hydrocarbons.
A paper by Zhu et al. estimated the maximum conversion efficiency of solar energy to biomass to be around 4-6%. [5] If, however, we simply focus on the conversion of photochemical energy into carbohydrates, then the efficiency increases to around 16% (6/[6+29+2]).
This last step could be thought of as the efficiency of converting electricity into green synthetic fuels and would give a cost ratio of 1:6, i.e. if 1J of electricity costs $1 then 1J of green synthetic fuel would cost $6. This compares to a current cost per joule ratio of 1:1. (Assuming a wholesale electricity price of $0.05 kwh-1 and 6 gigajoules of energy per $80 barrel of oil.) A 6 fold increase in the cost of gasoline relative to electricity would probably be sufficient to convert almost everyone to electric cars � especially if the cost of electricity itself has risen from its present day level.
Clearly the analysis above is much simplified. Whilst there is an implicit allowance for infrastructure costs (the plant needs to survive as well as just creating biomass), we have ignored any potential improvements to the biological method of carbohydrate synthesis. However the starting point for this work was the maximum biological conversion factor, and even a doubling of the efficiency would still imply a trebling in the cost of oil relative to the cost of electricity.
Liquid fuels occupy a central role in the world's transport system and, for some applications, are irreplaceable. As oil prices rise, synthetic fuels are likely to become more common. Unfortunately, absent any environmental legislation, these synthetic fuels are likely to be derived from alternative hydrocarbon stocks such as gas or coal. In the long run we will likely try to avoid using liquid fuels for all but the most critical applications.
© Adam Jorna. The author grants permission to copy, distribute and display this work in unaltered form, with attribution to the author, for noncommercial purposes only. All other rights, including commercial rights, are reserved to the author.
[1] S. Trasatti, "1799-1999: Alessandro Volta's 'Electric Pile' - Two Hundred Years, But It Doesn’t Seem Like It," J. Electroanal. Chem. 460, 1 (1999).
[2] A. Andrews and J. Logan, "Fischer-Tropsch Fuels from Coal, Natural Gas, and Biomass: Background and Policy," Congressional Research Service, RL34133, March 2008.
[3] F. Trott, Petrol 'Produced From Air and Water'," BBC News, 19 Oct 12.
[4] "BP Statistical Review of World Energy 2012," British Petroleum, June 2012, p. 20.
[5] M. Peel, "Shell GTL: Masterstroke or a Wrong Turn" Financial Times, 16 Dec 11.
[6] X.-G. Zhu, S. P. Long and D. R. Ort, "What Is the Maximum Efficiency With Which Photosynthesis Can Convert Solar Energy into Biomass?" Curr. Opinion Biotechnol. 19, 153 (2008).