 |
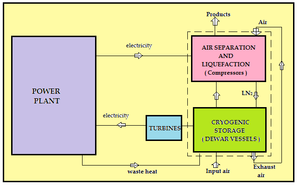
| Fig. 1: Schematic diagram of CES system. [5] |
The energy requirement of a region varies with time during the day and its characteristics also change with weather. Generally the demand for electricity is higher during the evening hours due to increased usage of lights. The power plant built for the region has to take care of this fluctuating load. A power plant is designed to have a capacity to fulfill the average demand for electricity. A power plant that is designed to produce a continuous constant supply of electricity that satisfies the average demand of the region is called base load power plant. Thus during the hours when the electricity demand is lower than the average demand, for example during early morning hours, excess energy is produced by the base load power plant and this energy is wasted. Peak loads hours are the hours when the demand is well above the average demands. During these hours, extra energy is needed to be supplied. Traditionally this is done by building a peak load power plant of a smaller capacity whose response time is less so that it can be switched on or off according to the demand fluctuation. However the major drawback of this system is that a lot of electricity is wasted when it is produced in excess of the required amount. If a system could be designed to store this excess energy produced during low demand hours and supplied during high demand hours, it would increase the efficiency of power plants greatly. This is especially important today because energy conservation has become a critical topic. One of the alternatives suggested to achieve this is cryogenic energy storage (CES).
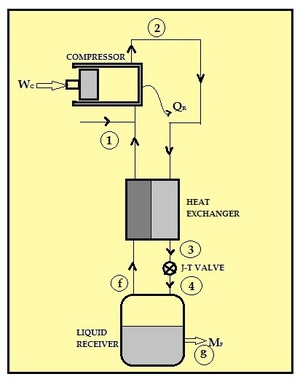 |
| Fig. 2: Nitrogen liquifaction by the Linde-Hampson Process. [2] |
Cryogenics is the science of producing extremely low temperatures ranging from 100K to absolute zero(0K). [1] At this temperature, the component gases of air like nitrogen, oxygen, hydrogen liquefy to produce cryogens. Cryogens exhibit properties that are very different from their corresponding gaseous forms. Other solid state objects like metals also exhibit alterations in properties when treated with cryogens. Due to this peculiarity, cryogens find extensive applications in the industry.
In a cryogenic energy storage system, excess energy produced by the power plant during off peak hours is used pull in the atmospheric air and compress it to produce cryogens, generally liquid nitrogen or oxygen. Temperatures as low as 77 K which is about the boiling point of nitrogen or lower have to be reached in order to liquefy air. During off peak hours, the stored cryogen is evaporated by environmental heat or heat from power plants. One of the problems faced in heat exchange at this point is that heat transfer coefficient on air side is very low. So a large amount of atmospheric air is required to bring about faster evaporation of the cryogen. If water is used as an alternative medium, a very large quantity of water is required. Thus thermal design of this system is a critical challenge in itself. Quick evaporation produces high pressure gases which are used to run turbines to produce electricity. At the same time, the gaseous oxygen can mixed with the combustion gases to enhance the process. Fig. 1 represents the schematic diagram of the parts of cryogenic energy storage system.
Air Liquefaction normally uses two processes: Linde-Hampson process and Claude process. In Linde-Hampson process, air from the atmosphere is compressed to a very high pressure of about 200 atm in a compressor. [2] This compressed air is passed through a counter-flow heat exchanger which reduces the temperature of compressed air. At high pressure and low cryogenic temperature, this compressed air is throttled in an insulated Joule-Thompson expansion valve to a low pressure. The liquid air is collected in storage tanks and the cold gaseous part is sent back through heat exchanger where it cools the incoming air to the compressor. The Claude process performs liquefaction in stages thereby reducing the pressure ratio requirements. The Claude process requires pressures of about 40 atm. [2]
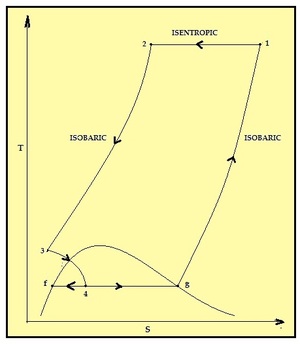 |
| Fig. 3: T-S diagram for the Linde-Hampson process. [2] |
The storage of the liquid nitrogen also presents substantial challenge. There are specialized flasks called Dewar vessels used to store liquid nitrogen. The Dewar vessel is a double walled container with high vacuum between the two walls. [3] An inlet and outlet is provided, the outlet being smaller than inlet. A vent is provided to allow the excess vapour formed due to heat leak to escape. This is a precautionary step to avoid excessive pressure build up inside the tank. The design of Dewar vessel should confirm to ASME Boiler and Pressure vessel code, sectionVIII(1983) for unfired vessels. [3]
CES has been successfully implemented at a power plant in Slough, UK by Highview Power Storage Ltd. The technology was originally developed by Peter Dearman for powering vehicles. [4] University of Leeds is also actively conducting research on this topic. Cryogenic energy storage is a green option because it uses air or nitrogen which is abundantly available in atmosphere and there are no direct emissions. More ever, if not for energy storage, the liquid air- Nitrogen or Oxygen- produced from the process can be used commercially or for refrigeration purposes. Cryogens have a huge application in metallurgical, medical, food, space etc. industries. New techniques of cryosurgery, blood preservation, cryo-treatment of metals are taking root. The drawback, however, of the process is that a very large amount of energy is required to compress the gases. So the efficiency of this process is very less (40-50%). [5] A lot of research is needed before this model can be standardized and commercialized.
© Chaitali Dalvi. The author grants permission to copy, distribute and display this work in unaltered form, with attribution to the author, for noncommercial purposes only. All other rights, including commercial rights, are reserved to the author.
[1] M. Mukhopadhyay, Fundamentals of Cryogenic Engineering (PHI Learning, 2010).
[2] R. F. Barron, Cryogenic Systems (Oxford, 1985).
[3] T. Flynn, Cryogenic Engineering, 2nd Ed (CRC Press, 2004).
[4] R. Harrabin, "Liquid Air 'Offers Energy Storage Hope'," BBC News, 1 Oct 12.
[5] H. Chen et al., "Progress in Electrical Energy Storage System: A Critical Review," Prog. Natural Sci. 19 291 (2009).