 |
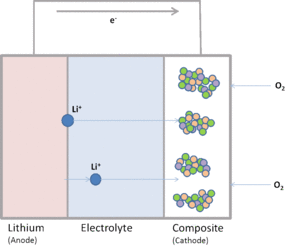
| Fig. 1: Schematic Li-air Battery |
The technological revolution over the past centuries consumes vast amount of energy and results in significant carbon footprint. A dominate proportion of overall energy demand is attributed to transportation sector which leads to various environmental problems, such as urban air pollution. Approximately 80% of national CO emission is accounted for transportations. [1] With foreseeable shortage of fuels and emphasis on being green, electrification of road transportation is a potential solution to both energy conservation and environmental protection. The advancement of electric cars has been sluggish in the past century due to the lack of suitable batteries. Lithium-ion batteries are generally considered the potent candidate for electric propulsion source in the near future. With an specific energy of 180 Wh/kg, though 5-fold higher than lead-acid batteries, a higher energy storage capacity is still desired to further reduce total battery weight on board and increase overall operation efficiency. [2] Li-air batteries, which is theoretically proved to be of high energy density, show a noticeable potential of being the future electric propulsion source with excellent carbon footprint record. [2]
The metal-air batteries are usually defined as batteries consist of metal-based anode and air-cathode which constantly extract oxygen from the ambient air. Specific batteries are characterized by the metal anode. For example, Li-air batteries refer to those containing lithium metal as anode material. The first metal-air battery is discovered by Leclanche in 1868. The nowadays commercialized zinc-air batteries is developed by Heise and Schumacher in 1932. Li-air batteries are first proposed in 1970s for their exceptionally high specific energy and power which qualify as a potential energy source for electric vehicle propulsion. [3] Non-aqueous Li-air batteries are first reported to be rechargeable by K.M. Abraham in 1996. The theoretical specific energy of Li-air batteries is calculated as 5,200 Wh/kg, or equivalently, 18.7 MJ/kg including oxygen. [4] Since oxygen is constantly drawn from air, specific energy is often quoted excluding oxygen content. This theoretical specific energy is calculated to be 11,140 Wh/kg, or 40.1MJ/kg which is close enough to that of gasoline, around 46MJ/kg. [4,5] Interest in Li-air batteries continues to grow because of the high energy capacity which is promising when comparing with gasoline. Due to engine inefficiencies, both gasoline and Li-air battery are predicted to achieve a practical specific energy of 1,700 Wh/kg which is several folds higher than most of the existing battery systems. [6]
A schematic basic non-aqueous Li-air battery cell, is illustrated in Fig. 1. The cell comprises a Li-based anode and an air cathode, contacted by non-aqueous electrolyte. The arrangement is similar to an aqueous Li-air battery. Porous carbon generally acts as cathode material supporting catalyst particles. Lithium in the anode undergoes a redox reaction, and lithium ions (Li+) are constantly transported through the electrolyte to the cathode and react with oxygen molecules. Lithium oxide (Li2O) and lithium peroxide (Li2O2) are generated in the air cathode. The general reaction are presented as: [7]
| Anode Reaction: | Li(s) ⇔ Li+ + e- | |
| Cathode Reaction: | Li+ + e- + 0.5 O2 ⇔ 0.5 Li2O2 | |
| Cathode Reaction: | Li+ + e- ⇔ 0.5 Li2O |
Despite the promising specific energy, Li-air batteries fall behind in several performance parameters. The highest reported achieved specific energy is 362 Wh/kg. [7] Although it is 100% higher than Li-ion batteries (~180Wh/kg), it only achieves 21% of the expected practical value. [8] The specific power is ~0.46mW/g which is only 10% of present Li-ion batteries and remains a major hurdle for electric vehicle propulsion. Life cycle is another important concern, as Li-air batteries degrade twice after first 50 cycles. [7] Development cycle of Li-air batteries is reviewed to be similar to that of Li-ion batteries. With essential parameters in automotive propulsion to be addressed in the future, the full transition to Li-air batteries might follow a similar path like Li-ion batteries which is 35 years of research and development. [9]
Although Li-air batteries present promising prospects of future road electrification, various limitations exist and remain major hurdles for this transition.
Li-air batteries are further categorized by electrolyte, namely, aqueous and non-aqueous. The estimated energy density is 1,300 Wh/kg for alkaline aqueous electrolyte and 1,400 Wh/kg for acidic aqueous electrolyte. [7] However, aqueous electrolyte contacts with Li anode and induces the redox reaction between lithium and water/acid, which speeds up the consumption of both lithium anode and electrolyte. Energy density of non-aqueous Li-air batteries is predicted to be 2,790 Wh/kg, and battery cells are terminated by air cathode being clogged by precipitated lithium oxides which are insoluble in electrolyte. [7] Recent research suggests that a combination of both types of electrolyte may provide a solution to both limitations, such as, adding additives to dissolve lithium oxides or compound electrolytes to reduce permeability between anode and electrolyte. The current energy density of Li-air batteries are noticeably lower than the values stated above, which is mainly resulted from the substantial weight percentage of electrolyte per cell (~70%) while both lithium and carbon content is only ~11%. [7]
Power density is an essential parameter is electric propulsion. Despite the high energy density, Li-air batteries are low in power density. During discharging process, oxygen is reduced to formed lithium-oxides, and the charging cycle reverses chemical reaction and produces oxygen gas. Both processes take place in the cathode surface. As a result, to ensure a satisfactory power output, a high surface area of cathode is substantial. [7] Rough estimation of a prototype Li-air battery shows that, with 100 kW power output and 1mA/cm2 current density at 2.5V requires an internal surface area of 4000 m2.
Li-air batteries fall short in round-trip efficiency which represents the ratio of energy discharged to energy needed during charging. Typical round-trip efficiency qualifying for electric propulsion is set at 90%. [9] However, the round-trip efficiency of Li-air batteries with pure carbon cathode is only 57%. [7] Although this record is further improved by cathode with platinum/gold catalysts (PtAu/C) to 73%, it is considered not realistic in commercialization of Li-air batteries since both platinum and gold are of extremely high price. [10]
Lifespan is another parameter when evaluating future potential of Li-air batteries as an electric propulsion source. Lithium oxides form during discharging cycle as lithium ions are transferred to the cathode and react with incoming oxygen. The recharging process involves the reduction of lithium oxides(Li2O and Li2O2). However, Li2O is not electrochemically active and subsequently not participating reversible reactions. The Li2O content varies from 0-100%, depending on types of electrolyte and carbon cathode. [7] The whole battery cell is terminated by accumulating lithium oxides clogging all pore volume of the cathode, since the clogging effect prevents further transports of both lithium ions and oxygen molecules.
Most research at present measure battery performance under high purity oxygen environment. In actual operation, as oxygen is extracted from ambient air, the moisture content in the air is also transported into the battery and reacts with lithium anode, which shortens battery lifespan. Recent research by Pacific Northwest National Laboratory (PNNL) employed membrane as a filtration interface enabling oxygen diffusion and moisture elimination, and operated in ambient condition with 21% oxygen and 20% of relative humidity. The results were encouraging, demonstrating an energy density of 362 Wh/kg over one month. [11]
Although Li-air batteries demonstrate an encouraging potential of being the future electric propulsion source, various limitations remain to be tackled before the full transition. With a promising high specific energy storage capacity, many challenges are to be overcome in the future research and development of Li-air batteries.
© Yuan Zhong. The author grants permission to copy, distribute and display this work in unaltered form, with attribution to the author, for noncommercial purposes only. All other rights, including commercial rights, are reserved to the author.
[1] "Our Nation's Air - Status and Trends Through 2008," U.S. Environmental Protection Agency, EPA-454/R-09-002, February 2010.
[2] M. Armand and J.-M. Tarascon, "Building Better Batteries," Nature, 451, pp 652 (2008).
[3] K. Kinoshita, Electrochemical Oxygen Technology (wiley, 192), pp 259-306.
[4] K. M. Abraham and Z. Jiang, "A Polymer Electrolyte-Based Rechargeable Lithium/Oxygen Battery," J. Electrochem. Soc. 143, 1 (1996).
[5] R. Schulz et al., "Recent Developments in the Applications of Nanocrystalline Materials to Hydrogen Technologies," Mat. Sci. Eng. A 267, 240 (1999).
[6] M. Jacoby, "Rechargeable Metal-Air Batteries," Chem. Eng. News, 88, 29 (2010).
[7] A. Kraytsberg and Y. Ein-Eli, "Review on Li-Air Batteries - Opportunities, Limitations and Perspective", J. Power Sources 196, 886 (2011).
[8] J. M. Tarascon, "Key Challenges in Future Li-Battery Research", Phil. Trans. Roy. Soc. A 368, 3227 (2010).
[9] G. Girishkumar et al., "Lithium-Air Battery: Promise and Challenges," J. Phys. Chem. Lett. 1 2193 (2010).
[10] Y. C. Lu et al., "Platinum-Gold Nanoparticles: A Highly Active Bifunctional Electrocatalyst for Rechargeable Lithium-Air Batteries," J. Am. Chem. Soc. 132, 12170 (2010).
[11] J.-G. Zhang et al. "Ambient Operation of Li/Air Batteries", J. Power Sources 195, 4332 (2010).