 |
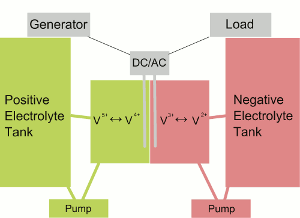
| Fig. 1: Flow Battery Diagram. Source: Wikimedia Commons |
With the rapid increase in production of intermittent energy sources such as wind and solar, there is an increasing need for large-scale electrical energy storage systems to more efficiently match supply and demand for these renewable sources. Also, large-scale energy storage can increase the annual load factor (defined as the annual mean power divided by the maximum three-day mean power) by load leveling. [1] Traditionally, pumped-hydro has been used for load leveling at large scale plants, but this is geographically limited to a small subset of locations.
Flow batteries are especially attractive for these leveling and stabilization applications for electric power companies. In addition, they are also useful for electric power customers such as factories and office buildings that require increased capacities, uninterrupted supply, or backup power. This paper will outline the basic concept of the flow battery and discuss current and potential applications with a focus on the vanadium chemistry.
A flow battery is a fully rechargeable electrical energy storage device where fluids containing the active materials are pumped through a cell, promoting reduction/oxidation on both sides of an ion-exchange membrane, resulting in an electrical potential. In a battery without bulk flow of the electrolyte, the electro-active material is stored internally in the electrodes. However, for flow batteries, the energy component is dissolved in the electrolyte itself. The electrolyte is stored in external tanks, usually one corresponding to the negative electrode and one to the positive electrode. The convention we will use is that the negative electrode is the anode and the positive electrode is the cathode when discharging.
The chemical process can be generalized to the following half reactions during discharge: [2]
| Anode Compartment: | An+1 - e- → An |
| Cathode Compartment: | Cn+1 + e- → Cn |
The charge neutrality condition for the each half-cell is maintained by a selective ion exchange membrane separating the anode and cathode compartments. The key differentiating factor of flow batteries is that the power and energy components are separate and can be scaled independently. The capacity is a function of the amount of electrolyte and concentration of the active ions, whereas the power is primarily a function of electrode area within the cell. Similar to lithium-ion cells, flow battery cells can be stacked in series to meet voltage requirements. However, the electrolyte tanks remain external to the system.
To optimize the efficiency of the cell, we can consider several related efficiencies, namely voltage efficiency, charge efficiency, power efficiency, and energy efficiency: [2]
| ηV = | Vdischarge Vcharge |
ηC = | Qdischarge Qcharge |
ηP = | IVdischarge IVcharge |
ηE = | Edischarge Echarge |
The efficiencies vary highly with the chemistry, state of charge, and process conditions, but the typical ranges are 62-73% voltage efficiency, 80-98% coulombic (charge) efficiency, and 66-75% energy efficiency. [2]
Energy density and power density are two of the most important characteristics of an energy storage system. Energy density is limited by the solubility of ions in the electrolyte solutions. Also, note that as the volume of the cell components gets small relative to the volume of the electrolytes, the flow battery approaches its theoretical maximum of energy density. Higher capacity systems are thus more efficient in this respect, as the majority of the weight is the electrolyte which directly stores energy. Since capacity is independent of the power-generating component, as in an internal combustion engine and gas tank, it can be increased by simple enlargement of the electrolyte storage tanks. Flow batteries allow for independent scaleup of power and capacity specifications since the chemical species are stored outside the cell.
The power each cell generates depends on the current density and voltage. Flow batteries have typically been operated at about 50 mA/cm2, approximately the same as batteries without convection. [3] However, material innovations in the electrodes and membrane have the potential to significantly reduce the internal resistance of the cell. Using a thinner membrane while maintaining ion selectivity has enabled some redox flow cells to achieve current densities as high as 80 mA/cm2 of electrode area in a 50 kW total output system. [4] However, as shown in Table 1 below, the overall power density still remains low compared to lead-acid and lithium-ion batteries since the volume encompassed by the aqueous reagents/products is so much higher. Thus, energy and power density are a primary weakness of flow batteries, and an area which future research has significant potential to improve.
|
||||||||||||||||||||||||||||||
| Table 1: Battery Comparison (based on data from [4]). The first five are flow batteries. | ||||||||||||||||||||||||||||||
Flow batteries, particularly those with reactions involving only valence changes of ions, are especially robust in their cycle lifetime, power loading, and charging rate. Since for non-hybrid flow batteries there are no concerns associated with solid active substances (such as with lithium-ion batteries, which experience significant degradation in capacity and efficiency over time), the electrolyte has an essentially indefinite lifetime and can be charged to full capacity and discharged completely with no adverse effects. The lifetime, limited by the battery stack components, is over 10,000 cycles for the vanadium flow battery. There is negligible loss of efficiency over its lifetime, and it can operate over a relatively wide temperature range.
The main benefits of flow batteries can be aggregated into a comprehensive value proposition. I will focus on the vanadium chemistry, as this has had the most commercial success. The main features that distinguish flow batteries are: [1]
Long service life: The semi-permanent electrolyte combined with minimal electrode degradation allows for a high number of full charge-discharge cycles before replacement is needed. The electrodes do not undergo physical/chemical changes, so they can be optimized for catalytic and electrical properties without having to design for holding active substances. Also, convective cooling of the electrodes by the pumped electrolyte aids in heat distribution and management.
No standby loss: During prolonged gaps in use, there is little self-discharge since the charge-carrying electrolyte is stored in separate tanks.
Low maintenance: The charge state of each cell is the same since the same electrolyte is used for all cells, thus overcharging is not necessary to guarantee a uniform a charge.
Recyclability & Safety: Waste vanadium can be reused and cross-contamination across the positive and negative electrode compartments does not affect the composition. Also, the electrolytes are relatively nontoxic.
Charging characteristics: Redox flow batteries are "not affected by fluctuating power demand, repeated total discharge, or charge rates as high as the maximum discharge rates." [2] These actions severely reduce cycle life in other batteries.
Modularity: Perhaps most important is that energy capacity can be scaled independently of the power; cell characteristics such as electrode area do not need to be changed to modify capacity. This allows for underground storage of electrolyte in freeform tanks, which has been demonstrated successfully in a 20 kW system. [1]
As discussed, these features lend themselves well to load leveling, voltage sag protection, and intermittent renewable sources integration applications. As enticing as the flow battery characteristics may seem, they must always be compared to alternative options such as lead-acid and lithium-ion batteries. The main detractor remains the low power and energy densities compared to other battery technologies. This is an integral problem with flow batteries, since charge density in solution is limited by solubility and is thus much lower than that possible in a nanostructured solid. The low energy and specific densities make flow batteries less suitable for portable applications where weight and volume are highly constrained.
However, there has been interest in potential electric vehicle applications, mostly due to the possibility of a nearly "instant recharge" by replacing the electrolyte at a charging station. A group at the University of Southampton investigated the possibility of using flow batteries in passenger vehicles. Their test vehicle was a modest hybrid electric with a top speed of 70 mph and electric range of 60 km (300 km extended range). [4] The 72 V, 110 Ah, 300 A lithium-ion battery used to achieve these specifications weighed 60 kg and occupied 96 L. For comparison, a flow battery with equivalent capacity and power would be 400 kg and have an estimated volume of 424 liters. [4] The group used characteristics of an optimized vanadium redox flow battery for its estimation. Clearly, the potential for EV applications is limited unless the energy density is greatly improved, or if there is a large cost differential compared to lithium-ion technology.
For the vanadium flow battery, vanadium metal actually comprises a majority of the cost. The price of vanadium is highly volatile. Cost analysis estimates that vanadium comprises approximately $50/kWh to $110/kWh of a total battery cost target of $100-200/kWh. [2] Companies such as Enervault claim to have reached this $100/kWh floor for large-scale energy storage. From a cost standpoint, a cheaper alternative to the vanadium redox couple with similar characteristics and higher solubility (resulting in higher energy density) would provide significant value.
Adoption of flow battery technology has been limited due to several technical and economic barriers; however there is a large and increasing market opportunity. As power generation becomes more and more decentralized with the advent of renewable sources, there will be a need to both increase load factors to efficiently meet peak demand as well as mitigate the intermittency of sources that lack predictable availability. These utility-scale applications will need energy storage in the megawatt range with a cycle life, rapid charge/discharge, and modularity that lead-acid is not optimized for. In the US, Enervault and Deeya Energy are private companies at the forefront of flow battery innovation and have attracted significant funding.
The energy density can be gradually improved through several incremental innovations. Three-dimensional, porous electrodes with optimized catalytic properties have the potential to significantly increase power output for a given electrode area. [2] Also, further research should be done in identifying stable redox couples that have high solubility in the desired temperature range while generating a high cell potential, especially if they are significantly cheaper than vanadium. [3] Further design engineering can also significantly improve performance, both at a cell level and system level. The goals are primarily to minimize pumping losses and minimize the costs of the membrane/electrode materials at the cell level, and provide the necessary control systems for grid integration and optimized power management at the system level.
Truly disruptive innovation entails an order of magnitude increase in both energy and power density. A critical limiting factor is the solubility of the electroactive species in the electrolyte solution. A spinoff of A123, known as 24M, plans to significantly increase the concentration of redox species by creating a flowing semisolid in contrast to a less viscous electrolyte. It plans to integrate the flow battery concept into the lithium-ion chemistry. The company applied for a patent in 2009 (US #20100047671) which details plans for a semisolid ion-storing electroactive material which is transported into and out of a cell assembly, similar to a flow battery architecture. It will have a preferable redox species concentration of at least 20 M, which if feasible will produce game-changing increases in energy density.
© Bhaskar Garg. The author grants permission to copy, distribute and display this work in unaltered form, with attribution to the author, for noncommercial purposes only. All other rights, including commercial rights, are reserved to the author.
[1] N. Tokuda et al., "Development of a Redox Flow Battery System," Sumimoto Electric Industries, SEI Technical Review 50, 88 (2000).
[2] C. Ponce De León, "Redox Flow Cells for Energy Conversion," J. Power Sources 160, 716 (2006).
[3] T. Nguyen and R. F. Savinell, "Flow Batteries," Interface 19, No. 3, 54 (2010).
[4] M. R. Mohamed, S. M. Sharkh and C. Walsh, "Redox Flow Batteries for Hybrid Electric Vehicles: Progress and Challenges," in IEEE Vehicle Power and Propulsion Conference VPPC '09, 7 Sep 09, p. 551.