Synfuel Cycle Efficiency
Darin Sleiter
October 24, 2010
Submitted as coursework for Physics 240,
Stanford University, Fall 2010
Introduction
A number of studies on the world oil and coal
reserves have stated that the world will use up all of the easily
accessible and reliable carbon-based fossil fuel sources in the next 150
years. [1-3] Carbon-based fuels support a majority of the current energy
economy, and it will likely take considerable time and money to build up
an infrastructure based on alternative energy sources to replace oil and
coal. However, even though the world will need to rethink where it gets
its energy, the way portable energy is stored and distributed in the
future for certain purposes does not necessarily need a complete
overhaul.
In particular, current car and truck transportation
is almost completely dependent upon gasoline and diesel fuels, and there
are serious obstacles that still need to be overcome for the leading
carbon-free alternatives, such as electric and hydrogen powered cars.
While these technologies are constantly being improved, gasoline still
has significant advantages over these alternate fuel sources in terms of
energy density, engine complexity, cost, safety, refuel times, and
sometimes even environmental impact. [4-6] It is possible that
alternative fuel sources will not surpass the performance of gasoline
before the world's oil resources are exhausted. Fortunately, the
technology already exists to allow the continued use of gasoline as an
energy storage medium for transportation, even after carbon-based fossil
fuels are exhausted.
In this paper, I will outline a renewable cycle for
creating synthetic gasoline out of CO2 extracted from the
atmosphere, and will discuss the technology and energy efficiency
associated with each of the four steps of the cycle. The first is the
extraction of carbon dioxide from the atmosphere. [7] The second and
third steps are the conversion of CO2 into gasoline using
methanol synthesis followed by the Exxon Mobil process. [8-10]. The
final step is combustion of the gasoline within a vehicle, releasing the
CO2 back into the atmosphere. The entire cycle is carbon
neutral since the carbon was initially extracted from the atmosphere or
the ocean. A similar process has been proposed in a white paper by
scientists at Los Alamos National Laboratory, where they call it Green
Freedom. [11]
CO2 Extraction
The basic idea of the CO2 extraction
presented in [7] is to first absorb CO2 from the atmosphere
into an aqueous solution, next to remove that solution from contact with
atmosphere into an enclosed chamber, and finally to extract the
CO2 from the solution and store it in a holding tank or pump
it directly to the methanol synthesis plant. The first step is made
possibly by using a chemical reaction that is strongly exothermic,
This reaction pulls CO2 from the air at a
rate roughly proportional to the concentration of carbon dioxide in the
air, and stores it in a liquid form where it can be then transported out
of the atmosphere. Next, within a separate enclosed chamber another
exothermic reaction forms a precipitate, removing the carbon from the
solution,
Next, this precipitate is dried and then heated in a
kiln in order to provide enough energy to drive the endothermic reaction
to release the CO2,
Once the CO2 is released, it is pumped
into either a holding tank, or directly into a methanol synthesis plant.
Following the release of all the CO2, the calcium is
converted back into a hydroxide for reuse by the application of steam in
a final reaction,
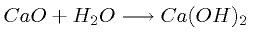 |
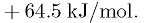 |
The sum of all the enthalpy changes is exactly zero,
however the process still requires energy. This is due to the
thermodynamic requirement that energy is used in order to reduce local
entropy by pulling the CO2 from the mixed atmosphere.
According to thermodynamics, the bare minimum energy requirement to
extract CO2 from the atmosphere is 19.5 kJ/mol. [7] The
physical process discussed in [7] actually requires much more energy
(442 kJ/mol) due to many necessary steps, such as actively blowing the
atmosphere across the solution in the first step, drying the lime
precipitate, and compressing the CO2. Even without
compressing or blowing the atmosphere, an energy consumption of 328
kJ/mol leads to a very low thermodynamic efficiency of 6%. However, more
CO2 is extracted from the atmosphere than is used to produce
the energy required to perform the extraction, and future improvements
could move the efficiency closer to the fundamental limit.
Conversion to Methanol
The next step in the process is to take the
compressed CO2 and convert it into Methanol. The net reaction
here is
While this reaction is exothermic, it is reversible,
and the opposite reaction will run at the same time and be more likely
at higher temperature. [12] Furthermore, the reaction rate is larger at
higher temperature, so an optimization of both high rate and low yield
is difficult. For this reason, a large amount of research has focused on
Cu/ZnO-based multicomponent catalysts [8,9] which can both increase the
yield and reaction rate. However, these catalysts can require
temperatures over 500 K to achieve substantial reaction rates.
Lower-temperature catalyst processes are being studied, but the problem
is far from optimized. [13] In order for this process to occur fast
enough and efficiently enough, the carbon dioxide conversion process
will require new catalysts pathways.
Neglecting the energy required to obtain the hydrogen
gas, we can approximate the energy cost of this process by assuming it
is required that the gas is heated to 500 K. The energy cost per mole
can be estimated as the energy required to heat the gas up from 300 K to
500 K at constant volume (0.028 kJ/(mol K) and 0.021 kJ/(mol K) for
CO2 and H2 , respectively) minus the enthalpy of
reaction. [14] Fortunately, the heat capacities of the reactants is so
small that this estimation results in a value of -31.3 kJ/mol, meaning
that the reaction produces more heat than is required to heat the gas to
500 K. If the system is designed to be continuous flow, no additional
heat input will be required to maintain a sufficiently high temperature.
We will include the energy cost for things during this conversion such
as pumping along with the conversion of methanol to gasoline in the next
step, assuming that both steps occur at the same plant.
Conversion to Gasoline
Purification of the resulting gas mixture before
feeding it into the Exxon Mobil methanol-to-gasoline (MTG) process is an
important step. However, the MTG process takes in a liquid mixture of
methanol and water, so a simple condensation step after the above
reaction would allow methanol and water to be removed from the gas
mixture. [10] The liquid mixture can then be pumped into the MTG process
while the remaining gas mixture is cycled for further CO2
conversion. We can assume that this condensation can be passive,
requiring no additional energy input.
In the MTG process, the main reaction is conversion
from methanol to hydrocarbon chains,
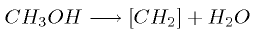
The length of the hydrocarbon chains produced is
dependent upon the actual process, but this process has been optimized
by Exxon Mobil and realized in a plant in New Zealand. [10]
The conversion to gasoline is exothermic, releasing
55.7 kJ/mol (1740 kJ/kg), and this energy is reused for pumping and
refining with a thermal efficiency of 54%. [10,15] Information about the
total energy required for the MTG processes is not easily accessible,
but we can attempt to estimate this energy requirement by looking at the
energy consumption of petroleum refineries in the US and comparing it to
the gasoline output of these refineries. In 2006, US refineries produced
3 billion barrels of finished motor gasoline. [16] At 42 gallons per
barrel and an average gasoline density of 720 kg/m3, this
corresponds to 3.4 × 1011 kg of gasoline produced. The
amount of energy consumed as a fuel (excluding feedstock) by US
refineries in 2006 was 3000 trillion btu, or 3.2 ×1015
kJ. [17,18] Dividing these values, we get 9400 kJ/kg as the average
energy requirement per kg of gasoline produced in a US refinery. This is
a very rough estimate, and is probably an over-estimate since US
petroleum refineries use energy to produce many other fuels besides
gasoline, while the MTG plant is streamlined exclusively for gasoline.
Therefore, it seems reasonable to assume that this estimate also covers
the energy for additional pumping and filtering required during the CO2
to methanol conversion.
If approximately half of the thermal heat produced by
the MTG process reactions is reused for refining as stated, then that
only accounts for approximately one tenth of the total energy
requirement. An additional 8500 kJ are required to convert
CO2 into gasoline for every kg of gasoline produced.
Combustion
The final step of the cycle is the combustion of the
gasoline within a vehicle, where the hydrocarbons react with oxygen to
create CO2 and water. The hydrocarbons in gasoline have an
average combustion energy of around 42000 kJ/kg. [19] This is the energy
content of the synthetic gasoline.
After combustion, the thermal energy released is
converted into motion. Engine thermal efficiencies are typically quoted
at around 35%, but researchers have obtained thermal efficiencies in a
lab averaging over 55%. [20] However, this efficiency will not be
included as part of the total synfuel conversion efficiency since it is
not related to the creation of the fuel.
Energy Conversion Efficiency
The total energy cost of extracting CO2
from the atmosphere and then converting it into gasoline can now be
computed. The extraction step takes 442 kJ/mol = 10000 kJ/kg, and the
conversion from CO2 to methanol to gasoline is estimated to
require 8500 kJ/kg. Comparing the energy stored in the gasoline,
Estored to the energy used during conversion,
Econv, we can compute the energy conversion efficiency
&\eta;,
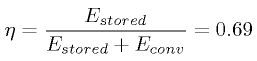
Thus 31% of the energy used to create the gasoline is
used during the conversion, placing a majority of the energy in the
gasoline. This efficiency does not take into account the H2
gas required in the conversion to methanol, which will require
additional energy to create or obtain. Note that the only net materials
consumed throughout the cycle are H2 gas during the methanol
step and O2 gas during the combustion, and the net product is
H20. Both of these can be obtained by electrolysis of water.
Conclusion
Gasoline usage will continue in the near future, and
possibly even after all the profitable carbon fossil fuels have been
used up. Gasoline is a very good portable storage medium for energy due
to its high energy density, ease of storage, and safety, among other
things. Here, we have considered a scenario where CO2 is
extracted from the air, and then converted into gasoline. We have
estimated the efficiency of the energy conversion to be 69%, which is
quite reasonable. However, the cost of gasoline obtained this way and
the throughput of the CO2 extraction or conversion steps have
not been considered here. The first will limit whether or not this
process is profitable, and the second will limit how widely used this
gasoline will be.
Furthermore, competing fuels and processes have not
been considered. Ethanol, diesel, methanol, syngas, and other
hydrocarbon fuels can all be manufactured with similar processes. Carbon
can also be extracted from plant material, such as corn stover, which
could prove to be a more efficient source of carbon. However, the
technology to create gasoline from CO2 is already available,
and if no other process or fuel is proven to be more reliable or
cheaper, gasoline fuel can be manufactured for transportation uses,
allowing the cars to continue running well after fossil fuels are
exhausted.
© Darin Sleiter. The author grants permission to
copy, distribute and display this work in unaltered form, with the
attribution to the author, for noncommercial purposes only. All other
rights, including commercial rights, are reserved to the author.
References
[1] BP Statistical Review of World Energy 2008,
British Petroleum, 2008.
[2] U. S. Energy Information Administration - World Estimated
Recoverable Coal - October 2008.
[3] S. Shafiee and E. Topal, "When Will Fossil Fuel
Reserves Be Diminished?," Energy Policy 37, 181 (2009).
[4] H. L. MacLean et al., "A Life-Cycle
Comparison of Alternative Automobile Fuels," Journal of the Air &
Waste Management Association 50, 1769 (2000).
[5] M. Ball and M. Wietschel, "The Future of Hydrogen
- Opportunities and Challenges," Intl. J. Hydrogen Energy 34, 615
(2009).
[6] J. Romm, "The Car and the Fuel of the Future,"
Energy Policy 34, 2609 (2006).
[7] F. Zeman, "Energy and material Balance of
CO2 Capture from Ambient Air," Environ. Sci. Technol.
41, 7558 (2007).
[8] M. Saito , "Development of
Copper/Zinc Oxide-Based Multicomponent Catalysts for Methanol Synthesis
from Carbon Dioxide and Hydrogen," Applied Catalysis A 138, 311
(1996).
[9] J. Toyir et al., "Highly Effective
Conversion of CO2 to Methanol Over Supported and Promoted
Copper-Based Catalysts: Influence of Support and Promoter," Applied
Catalysis B 29, 207 (2001).
[10] F. J. Keil, "Methanol-to-Hydrocarbons: Process
Technology," Microporous and Mesoporous Materials 29, 49
(1999).
[11] F. J. Martin and W. L. Kubic, "Green Freedom, A
Concept for Producing Carbon-Neutral Synthetic Fuels and Chemicals," Los
Alamos National Laboratory,
LA-UR-07-7897,
November 2007.
[12] X. Liu et al., "Recent Advances in
Catalysts for Methanol Synthesis via Hydrogenation of CO and
CO2", Ind. Eng. Chem. Res. 42, 6518 (2003).
[13] N. Tsubaki, M. Ito and K. Fujimoto, "A New
Method of Low-Temperature Methanol Synthesis", Journal of Catalysis
197, 224 (2001).
[14] "
Gas Encyclopedia," Air Liquide.
[15] C. J. Maiden, "The New Zealand Gas-to-Gasoline
Project", Studies in Surface Science and Catalysis 36, 1
(1988).
[16] "Topics for
Petroleum Refining and Processing," U.S. Energy Information
Administration.
[17] "Safety Data Sheet: BP Autogas," British
Petroleum,
SUK219,
1 Dec 02.
[18] "Manufacturing
Energy Consumption Survey," U.S. Energy Information
Administration.
[19] V.T. Bugai, A.V. Oreshenkov, and O.A.
Burmistrov, "Determination of Heat of Combustion of Fuels by
Calculation", Chemistry and Technology of Fuels and Oils, 34, 5
(1998).
[20] J. Kunanoppadon, "Thermal Efficiency of a
Combined Turbocharger Set with Gasoline Engine", Am. J. Eng. Appl. Sci.
3, 342 (2010).