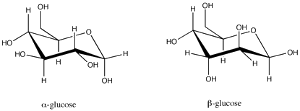
 |
| Fig. 1: Structures of α- and β-glucose. |
Biofuel is considered as one of the alternative energy sources in the near future when oil and coal are gone. Currently the major feedstock of biofuel is sugar from starch, while little comes from lignocellulosic biomass because of cost. However, the majority of the total carbohydrates in biomass is presented in forms of lignocelluloses like cellulose, hemicellulose or lignin. [1] A lot of biomass cannot be utilized if there is no cheap way to break down cellulose. In this article, we will focus on the structure of cellulose, cellulose hydrolysis techniques, current research of enzymatic hydrolysis and the cost of enzymatic hydrolysis.
D-glucose is the only building block of many polysaccharides like glycogen, starch and cellulose. Glucose mostly exists in the more stable hemiacetal form, which has two isomers: α- and β-glucose. (Fig. 1) Glycogen and starch are branched polymers consisting α-glucose, while cellulose molecules are unbranched chains formed by β-glucose. A basic repeating unit of cellulose contains two β-glucose molecules connected via a ketal linkage, with one β-glucose flipped by 180 degrees, as shown in Fig. 2. One cellulose molecule normally consists of a few hundreds to thousands of glucoses. The unbranched cellulose chains are very densely packed via inter-chain hydrogen bonds. Benefitted from the dense packing, celluloses form strong supports for plants.
Hydrolysis of cellulose is very critical for biofuel production, because only glucose, not cellulose, can be consumed by the bacteria used in fermentation to produce biofuel. Why is the hydrolysis of cellulose difficult? Celluloses have crystalline structures due to the dense packing of cellulose chains. They are very stable under many chemical conditions. They are not soluble in water, many organic solvents, weak acids or bases. The crystalline structure can be destroyed and turned into amorphous form under high temperature (>300 C) and pressure (25 MPa). [2] There are normally two ways to hydrolyze cellulose: chemically and enzymatically. The chemical method is to use concentrated strong acids to hydrolyze cellulose under high temperature and pressure. [3] However, this method is not preferred by biofuel industry, because toxic byproducts remaining in the glucose products will be introduced into the fermentation step, affecting the fermenting bacteria/yeast. Hence, the milder enzymatic method seems to be a much more potential candidate to hydrolyze cellulose.
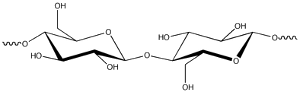 |
| Fig. 2: Cellulose subunit. |
The enzymatic method uses bacteria secreted proteins to hydrolyze cellulose. This method involves a lot of enzymes, namely cellulase. Research has shown that these enzymes play different roles cooperatively in the hydrolysis of cellulose: some cleave the cellulose chain from the middle into fragments containing 4-5 glucoses, some breakdown these fragments into smaller units of two glucoses, and some finally turn these small units into single glucose. [4] However, the research of enzymatic hydrolysis is still at the early stage. There are many important questions remain unanswered, such as the number of critical enzymes needed for the hydrolysis, the crystal structures of the enzymes, the exact mechanisms of hydrolysis, etc. In the next 10-20 years, the research in this area is expected to make great progresses, therefore a lot of protein engineering work will be accomplished to enhance the efficiency and lower the cost of the hydrolysis of cellulose.
It is much more expensive to obtain biofuel from cellulose than from starch. The cost of ethanol produced form cellulose is around $1.5 per gallon, where as that produced from corn starch is around $1 per gallon. [5] Hydrolysis of cellulose becomes the bottleneck of bringing down the cost of biofuel production from cellulose. About half of the total cost of producing biofuel from cellulose is allocated on enzymatic cellulose hydrolysis, in which cellulase is the most expensive part, consuming 15-25% of the total cost. [6] Enzymatic hydrolysis is a very slow step. As a result, a lot of cellulases are needed to achieve a reasonable hydrolyzing rate. Typically, the ratio of cellulase to cellulose used in hydrolysis step is 25 g/1 kg. [7] Despite that the fermentation can produce a great amount of cellulase, about 100 g from 1liter broth, the cost of cellulase still remains a large portion of the total cost.
In summary, the molecular structure of cellulose makes the hydrolysis of cellulose very difficult and costly. Our knowledge on the hydrolysis of cellulose is far away from sufficient to allow us producing biofuel with a high efficiency and low cost. However, if an effective and cheap cellulase can be developed, we will be able to obtain biofuel at a much lower price and utilize more parts of the plants.
© Xi Jin. The author grants permission to copy, distribute and display this work in unaltered form, with attribution to the author, for noncommercial purposes only. All other rights, including commercial rights, are reserved to the author.
[1] B. R. Hames, "Biomass Compositional Analysis for Energy Applications," Methods Mol. Biol. 581, 145 (2009).
[2] S. Deguchi, K. Tsujii and K. Horikoshi, "Cooking Cellulose in Hot and Compressed Water," Chem. Commun. 61, 3293 (2006).
[3] The Future of Ethanol" BlueFire Ethanol.
[4] R. Kumar, S. Singh, and O. V. Singh, "Bioconversion of Lignocellulosic Biomass: Biochemical and Molecular Perspectives," J. Ind. Microbiol. Biotechnol. 35, 377 (2008).
[5] R. Wallace et al., "Feasibility Study for Co-Locating and Integrating Ethanol Production Plants from Corn Starch and Lignocellulosic Feedstocks," U.S. National Renewable Energy Laboratory, NREL/TP-510-37092, January 2005.
[6] L. R. Lynd et al., "How Biotech Can Transform Biofuels," Nat. Biotechnol. 26, 169 (2008).
[7] A. Carroll and C. Somerville, "Cellulosic Biofuels," Annu. Rev. Plant Biol. 60, 165 (2009).