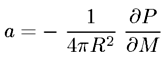
|
||||||||||||
| Table 1: Global Warming Potentials for Common Greenhouse Gases [2] |
In 1978, the United States federal government banned the use of Chlorofluorocarbons (CFCs) in aerosol cans. The cited reasons had to do with the extremely detrimental effect that the emissions of these gases had on the depletion of ozone in the atmosphere. [1] Ozone serves as a buffer that effectively limits the amount of UV rays entering the lower levels of the atmosphere which we as humans occupy. Without the ozone being present, we would have significantly higher levels of skin cancer and other simpler species would easily die. CFCs have chlorine molecules in them. Upon entering the atmosphere where significant amounts of ozone exist, the chlorine molecules become free and react with the ozone in the atmosphere thus increasing the amount of oxygen, but reducing the ozone present. This reduction of ozone in the atmosphere can have significant impacts on human life that could accelerate global warming at a significantly higher rate. In short, CFCs have a huge impact on the atmosphere when emitted. To quantify this impact, greenhouse gas equivalency factors can be used to equate the environmental impact of different gases.
Global warming potential (GWP) is a relative measure of how much a particular gas contributes to global warming. The baseline metric for global warming potential is one carbon dioxide molecule. Consequently, carbon dioxide has a GWP of 1. When dealing with carbon dioxide, the amount of molecules being emitted is the only metric that needs to be quantified in order to come to a GWP approximation. However, if one were to try and come to an equivalent GWP number for a greenhouse gas other than carbon dioxide, such as CFCs, a greenhouse gas equivalency factor would need to be used. In the case of CFCs, this equivalency factor ranges from 400 to 15,000 depending on the time horizon. Why is there such a large variation for CFCs' equivalency factors and why do they differ depending on a time horizon?
The Global Warming Potential GWP is a time-normalized thermal resistance index. The radiative efficiency of a gas is [3]
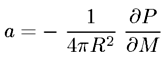
where P is the radiative power transmitted through the troposphere-stratosphere boundary, R is the radius of the earth, and M is the mass of this gas added to the atmosphere, with the temperature profile of the troposphere held fixed. The GWP of chemical species x is then given by [3]
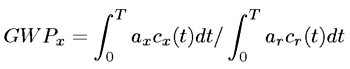
where c(t) is the amount of 1 kg of the gas injected at t = 0 and still in the air at time t, , T is the time horizon and r denotes a reference species, typically carbon dioxide. The forcing capacity is a function of the infrared absorbance, path length of the gas in question and density. GWP then adjusts this capacity according to how long it would take for the gas to naturally decay in the atmosphere. A gas that takes longer to decay naturally is more harmful than one that can decay in a short time span. Once gases start accumulating at a faster rate than they are able to decay, global warming runs away. The GWP is heavily dependent on the time horizon being used. Table 1 assumes a 100-year time horizon, which is the standard time horizon used for citing GWP values. The next section details the importance of time horizon.
Common values used for calculating the time horizon include 25, 50 and 100 year timespans. US government reports often use 100 years as the assumed baseline for this metric. For example, in the table above the emission of one molecule of methane would be the equivalent of 25 molecules of carbon dioxide. It would take 100 years for this one molecule of methane to naturally dissolve in the atmosphere at the 25 molecule carbon dioxide equivalency rate. Similarly, for a 20 year time horizon, the equivalency rate would be 72 molecules of carbon dioxide for one molecule of methane. Consequently, as the time horizon goes up, the equivalency rate goes down and as the time horizon decreases, the equivalency rate increases. Given the complexities of atmospheric science, this is not necessarily a linear relationship and detailed mathematical models are developed in order to understand this exact correlation.
With the BWP metric standardized it becomes possible to convert greenhouase gases into equivalents of carbon dioxide or carbon. The distinction between carbon dioxide and carbon is sometimes confusing. Carbon Dioxide is a three atom molecule with a molar mass of 44 grams whereas carbon is a single atom with a molar mass of 12 grams. [4,5] In order to convert between carbon dioxide equivalents and carbon equivalents, these molar mass differences need to be accounted for through the simple application of appropriate stoichiometric relationships. Most modern literature use carbon dioxide equivalents, but older sources may use carbon equivalents.
As stated in the beginning, CFCs were banned due to the fact that they caused significant ozone depletion when released into the atmosphere. However, carbon dioxide has a different mechanism for increasing the Earth's temperature. This involves trapping the heat produced from the Earth inside the atmosphere thus driving up the temperature of the planet. Given the fact that depleting ozone could have significantly more catastrophic effects than the accumulation of carbon dioxide in the atmosphere, it sounds reasonable that CFCs have such a high GWP. Carbon dioxide and other greenhouse gases both have a part to play with regard to climate change. Understanding which ones have the largest impact is critical to creating a method to manage emissions that could have the biggest impact and mitigate the effects of climate change.
copy; Subhan Ali. The author grants permission to copy, distribute and display this work in unaltered form, with attribution to the author, for noncommercial purposes only. All other rights, including commercial rights, are reserved to the author.
[1] L. E. Manzer, "The CFC-Ozone Issue: Progress on the Development of Alternatives to CFCs," Science 249, 31 (1990).
[2] "IPCC Fourth Assessment Report: Climate Change 2007 (AR4)," Intergovernment Panel on Climate Change.
[3] "IPCC Third Assessment Report: Climate Change 2001 (TAR)," Intergovernment Panel on Climate Change.
[4] "Metrics for Expressing Greenhouse Gas Emissions: Carbon Equivalents and Carbon Dioxide Equivalents," Environmental Protection Agency, EPA420-F-05-002, February 2005.
[5] U. Springer, "The Market for Tradable GHG Permits Under the Kyoto Protocol: a Survey of Model Studies," Energy Economics 25, 527 (2003).