The discovery of nuclear fission is perhaps one of the most influential moments in the history of science, the source of both nuclear energy and nuclear weapons. Historically however, its discovery was quite an accident, a side effect from physicists and chemists naively exploring the internal components of the atom in more and more detail. At the start of the 20th century, physicists started to view the atom as made up of even smaller subatomic particles. In 1887 J. J. Thompson discovered the electron. In 1911, Rutherford's famous gold-foil experiment confirmed the existence on a positively charged nucleus. The discovery of the neutron in 1932 completed the modern basic picture of the atom and was the key to unlocking the power of fission.
The first notion of a neutral subatomic particle can be traced to as early as 1920. Ernst Rutherford in his Bakerian Lecture hypothesized over a hybrid particle consisting of a negatively charged electron bound tightly to a positively charged proton, like a hydrogen atom but with the electron located basically inside the nucleus. [1] The charges could cancel each other out creating a neutron particle with approximately the mass of a proton. Rutherford in his lecture had already begun to speculate the properties of such a particle, the most relevant to fission being that with no electrical charge, the "neutron" as he called it, would be able to enter the positively charged nucleus. This key feature is exactly what makes the future discovery of fission possible; in fact, fission was discovered just 7 years after the discovery of the neutron.
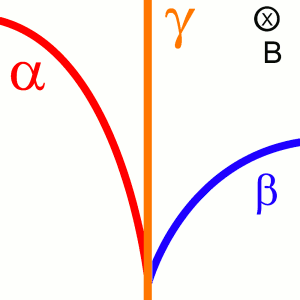
German physicists Walther Bothe and Herbert Becker discovered in 1930 that Beryllium emitted a seemingly ultra high power neutral radiation when bombarded by alpha particles from a polonium source. Up until then, there were three known types of radiation named alpha, beta, and gamma. The first two types are charged helium nuclei and electrons respectively and therefore were bent by magnetic fields. The third, gamma radiation, was known to be high energy photons and hence were not deflected by magnetic fields. Bothe and Becker therefore concluded that Beryllium must emit a sort of high energy gamma ray. However, there was one discerning feature between this mysterious radiation and gamma rays that remained: the mystery radiation was able to penetrate up to 20 cm of lead. By contrast, gamma rays at the energies commonly encountered were stopped by less than a couple of millimeters. Bothe and Becker wrote this discrepancy off by assuming that these gammas were of an ultra high energy. [2]
However, James Chadwick, a previous student of Rutherford, was not convinced. Previous experiments by the Curie-Joliot’s had measured the energy of this radiation to be 50 MeV. By placing a hydrogen rich paraffin material in the path of the radiation, the Curie-Joliot’s were able to observe protons ejected from the paraffin. By measuring the range of such protons in air to be 26 cm, they were able to determine the speed of the ejected protons to be at max 3 × 109 cm/s. Assuming a Compton scattering like interaction between the proposed high energy gamma and the protons in the paraffin, they arrived at the calculated photon energy of around 50 MeV. [3]
Chadwick was deeply disturbed by this result and repeated the experiment using slightly more accurate apparatus but arrived at a similar energy of 52 MeV. In February of 1932, Chadwick wrote a letter to the editor of Nature describing his results and determined that the quantum theory of light could only account for up to 14 MeV of the energy transferred while still obeying conservation of energy and momentum. [4] The other 38 MeV were still unaccounted for if this radiation was a high energy proton. Chadwick goes further in his letter to speculate that these penetrating effects could be explained by a "neutron" like the one proposed back in 1920 by Rutherford.
Chadwick decided to probe deeper into this excited Beryllium radiation. He discovered that this radiation not only ejected protons, but also other light elements. Describing this phenomenon as a type of photon radiation became even more implausible as ejecting heavier particles required more energy than the protons in the paraffin. It was clear to Chadwick that either conservation of energy and momentum must be thrown away, or that a new hypothesis of what this radiation was must be formed. He asserts that by assuming the radiation was a neutral particle with roughly the mass of a proton. Energy transfer between two colliding particles is most efficient when the two masses are on the same order (See here for a calculation why). Therefore, by making the radiation particle’s mass the same as a proton, it became easy to explain how the paraffin’s protons were ejected with such speed.
The radiation’s deeply penetrating power could also be easily explained due to its neutral charge. Since the neutron lacks electric charge, it does not interact easily through the electric magnetic force. As a result, the positively charged nuclei of matter would not repel or deflect neutrons as easily as they did with alpha particles or even gamma rays. By comparing measurements for the maximum velocity these neutrons could impart to hydrogen and nitrogen, Chadwick was able to prove that the mass of this neutral particle was indeed 1.15 ± 0.1 the massof the proton. [5] Chadwick had finally discovered the elusive neutron.
Following the discovery of the neutron, it did not take very long for physicists to begin experiments exploring its power. In 1934, Enrico Fermi decided to try to create new radioactivity by bombarding different elements with neutrons. [6] He discovered that elements above fluorine were activated radiated with a neutron source. Fermi concluded that these elements, including Uranium, were either transformed into a new isotope of the same element, or one a couple of atomic numbers lower. However, shortly after his experiment was published, Ida Noddack, an organic chemist from Berlin challenged Fermi's chemical separations and proposed that perhaps Uranium was splitting into two or more large fragments instead. [7] It seemed inconceivable to Fermi and physicists of the time that the nucleus wasn’t a stable unit.
Between 1935 and 1938 German physicists Otto Hahn, and Fritz Strassmann discovered that there were additional products produced from Uranium bombarded by neutrons. [8] This is where the great accidental discovery of fission occurs. Using chemical analysis, Hahn and Strassmann initially assumed that one of these products was Radium, four elements below Uranium. They decided to isolate this "Radium" product using a Barium catalyst. A catalyst is a chemical added to speed up a reaction, and then removed unchanged at the end. However, when Hahn and Strassmann tried to remove the Barium catalyst, they found that their solution was inseparable! Both Radium and Barium are group II elements and therefore have similar chemical properties, but Barium is almost a whole 40 elements below Uranium so it was never considered as a possible product.
Hahn and Strassmann communicated these results to another pair of German physicists Lise Meitner and her nephew Otto Robert Frisch. Using Bohr’s model of the nucleus as a water drop, Meitner and Frisch formulated a new theory of how neutrons affect Uranium. [9] They proposed that instead of just chipping off a few protons, the incoming neutron could split the Uranium nucleus into two large fragments of comparable size, including one of the element Barium as observed above. Like a large water droplet, adding violent movement can sometimes cause the droplet to divide into two. Frisch named this new nuclear process fission after the term for how biological cells divide. From this accidental discovery by two chemists, bred by the ignorance of physicists including Fermi, nuclear fission has enabled both energy sources and violent destruction far surpassing anything before it.
© 2009 Michael Liu. The author grants permission to copy, distribute and display this work in unaltered form, with attribution to the author, for noncommercial purposes only. All other rights, including commercial rights, are reserved to the author.
[1] E. Rutherford, "Nuclear Constitution of Atoms," Proc. Roy. Soc. A 97, 374 (1920).
[2] Bothe and H. Becker, "Küstliche Erreichung von Kern-Strahlen," Z. Phys. 66, 289 (1930).
[3] I. Curie, C.R. Acad. Sci. Paris, 193,1412 (1931).
[4] J. Chadwick, "Possible Existence of a Neutron," Nature 129, 312 (1932).
[5] J. Chadwick, “The Existence of a Neutron,” Proc. Roy. Soc. A 136, 692 (1932).
[6]E. Fermi, "Artificial Radioactivity Produced by Neutron Bombardment," Proc. Roy. Soc., A 146, 483 (1934).
[7] I. Noddack, "Über das Element 93," Zeitschrift für Angewandte Chemie 47, 653 (1934).
[8] O. Hahn and F. Strassmann, "Concerning the Existence of Alkaline Earth Metals Resulting from Neutron Irradiation of Uranium," Naturwissenschaften 27, 11 (1939).
[9] O. Frisch and L. Meitner, "Disintegration of Uranium by Neutrons: A New Type of Nuclear Reaction," Nature 143 239 (1939).