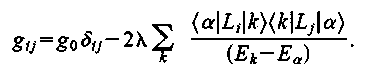
Electron Spin/Paramagnetic Resonance (ESR or EPR) is a versatile and non-destructive spectroscopic technique which is used to detect species that are paramagnetic or have unpaired electrons. It deals with the interaction of electromagnetic radiation with the intrinsic spin magnetic moment of electrons. Over the past 50 years, ESR has been applied to some of the following fields of research:
A. Physics
B. Chemistry
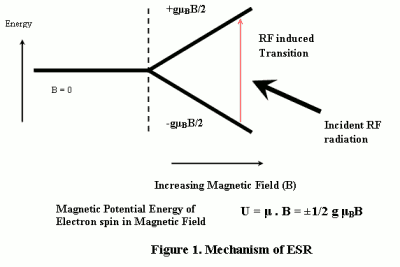
Transitions can be induced between unpaired electron spin states, by applying a magnetic field (B) and then supplying electromagnetic energy (usually in the microwave range of frequencies). [Figure 1].
An electron has a magnetic moment associated with the spin. The nature of an isolated electron spin is such that two and only two orientations are possible. As per the Zeeman effect, the application of an external magnetic field (B) (e.g 0.34 Tesla) provides magnetic potential energy which splits the spin states by an amount proportional to the magnetic field. The electromagnetic radiation (~10 GHz corresponding to 0.34 Tesla magnetic field) can then cause a transition from one spin state to the other. The energy associated with this transition is expressed in terms of B, Lande factor g and Bohr magneton [1] (See Equation in Figure 1).
A free electron has a 'g' value of 2.002319304386. Deviations from the isolated electron case arise in real solids due to:
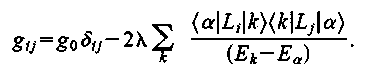
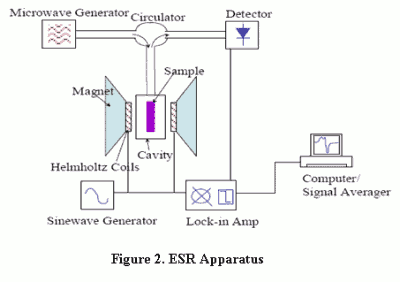
ESR signals/spectra can be generated by resonant energy absorption measurements made by varying the magnetic field B and using a constant frequency radiation. The ESR measurement apparatus [Figure 2] consists of:
The Si-SiO2 is the most important interface in the Metal Oxide Semiconductor Field Effect Transistor (MOSFET), which has become the dominant device for Very Large Scale Integrated Circuits (VLSI). A microprocessor or a mobile phone has > 50 million of these devices and interfaces. The performance and reliability of a MOS device is heavily influenced by the quality and properties of this interface.
 |
 |
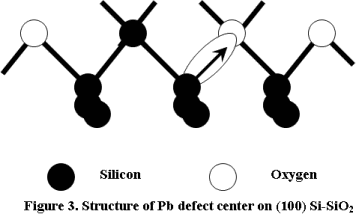
A major effort on ESR studies of point defects in bulk Silicon [2] had begun at about the same time as the MOS research and development. The premier work of Watkins et al demonstrated an "intrinsic" defect - the Silicon divacancy and its direct production by high energy electron irradiation. Later on the MOS Si-SiO2 interface had been shown to dominate the device properties. Hence a lot of research effort went into characterizing this interface. Nishi et al identified a paramagnetic defect called the Pb center [3] (Figure 3) as trivalent silicon (popularly referred as silicon “dangling bond”) at the Si/SiO2 interface. They detected three ESR signals from (111) Silicon at g=2.000 to 2.010 and g=2.06 to 2.07. These were termed as the Pa, Pb and Pc, respectively. They were assigned to trapped electrons in the oxide (Pa), interface trivalent Silicon centers (Pb) and trapped holes in the Silicon (Pc).
On the (100) surface, two tilted (111) orientations were observed. In addition to this a chemically different center was seen in two beveled orientations, which was named Pb1. Following this discovery the original trivalent interface defect center was re-named Pb0.
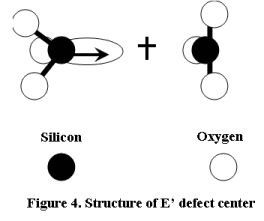
Other defects in the MOS oxide that have been identified are E' states [4], which are usually holes trapped at oxygen vacancies (Figure 4) or an unpaired electron localized on a silicon backboned to three oxygen atoms.
The ESR g value of an axially symmetric defect [6] (most of the Si-SiO2 defects are axial or have near axial symmetry) is given by:
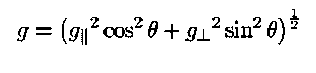
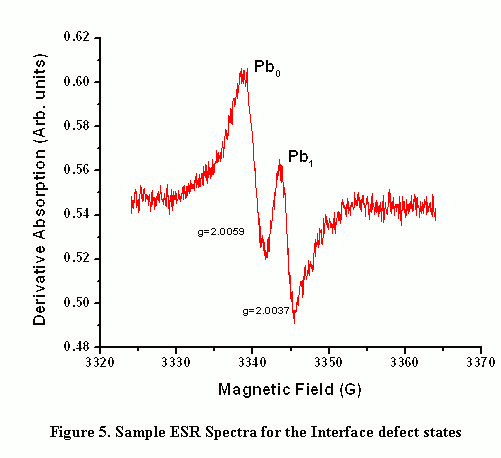
where g|| corresponds to the tensor value for the magnetic field parallel to the defect axis of symmetry. A sample ESR spectrum (obtained by the author in Nishi ESR lab, Electrical Engineering Department, Stanford University) is shown in Figure 5. By making comparisons with calibrated spin standards of Pb0 and Pb1 and spectrum fitting - the "g" values and amount of defect densities can be determined.
Current research is focused on comparing Plasma and Thermal Oxides using ESR. (This work has not been included in this report.)
© 2007 Gaurav Thareja. The author grants permission to copy, distribute and display this work in unaltered form, with attribution to the author, for noncommercial purposes only. All other rights, including commercial rights, are reserved to the author.
[1] J.E. Wertz and J.R. Bolton, ESR (Chapman and Hall, 1986).
[2] G.D. Watkins and J.W.Corbett, "Defects in Irradiated Silicon I: Electron Spin Resonance of the Si-A Center," Phys. Rev. 121, 1001 (1961).
[3] Y. Nishi, Jpn. J. Appl. Phys. 10, 53 (1971).
[4] E. H. Poindexter, Semicond. Sci. Tech. 4, 961 (1989).
[5] P.M. Lenahan, and J.F. Conley, J. Vac. Sci. Tech. B 16, 2134 (1998).
[6] P.M. Lenahan, and J.F. Conley, Trans. Dev. Mat. Rel. 5, 90 (2005).