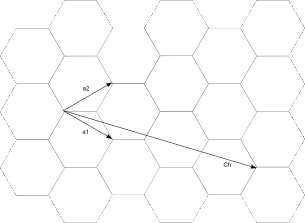
 |
| Fig. 1: Unit cell vectors of graphene, a1 and a2. The vector Ch = n1a1 + n2a2 defines the circumference of the nanotube. |
Carbon nanotubes have great potential in the future of nanoscale electronic devices - whether as 1D nanowires or as components in transistors. To understand why some nanotubes behave as metals and others as semiconductors, one must understand the band structure of nanotubes. By modifying the boundary conditions for an infinite graphene sheet, the electronic behavior of carbon nanotubes can be predicted as a function of chirality. To utilize this information, it is necessary to be able to isolate metallic or semiconducting carbon nanotubes, depending on the intended application. This report will briefly discuss the calculations, measurements, and separation techniques pertaining to the electronic properties of carbon nanotubes.
To understand how to perform a tight-binding calculation on a carbon nanotubes, one should begin with an infinite graphene sheet. These two forms of carbon share an atomic arrangement and thus, some of their electronic properties are expected to be similar. If we consider the two-atom basis of graphene, we can define vectors d1, d2, and d3 to be the nearest-neighbor vectors from the atom at the origin of the unit cell to the three atoms surrounding it. Additionally, let us define the following vectors:
| g0 = eik·d1 + eik·d2 + eik·d3 | g1 = √3/2(eik·d1 - eik·d2) |
| g2 = 1/2eik·d1 + 1/2eik·d2 - eik·d3 | g3 = √3/2(eik·d1 + eik·d2) |
| g4 = 1/4eik·d1 + 1/4eik·d2 + eik·d3 |
With these and the Harrison universal parameter definitions, an orthogonal tight binding calculation on graphene requires diagonalizing the following 8x8 matrix:
| εs | 0 | 0 | 0 | Vssg0 | Vspg1 | Vspg2 | 0 |
| 0 | εp | 0 | 0 | -Vspg1 | Vppσg3 + Vppπg4 | 1/2(Vppσ - Vppπ)g1 | 0 |
| 0 | 0 | εp | 0 | -Vspg2 | 1/2(Vppσ - Vppπ)g1 | Vppσg4 + Vppπg3 | 0 |
| 0 | 0 | 0 | εp | 0 | 0 | 0 | Vppπg0* |
| Vssg0* | -Vspg1* | -Vspg2* | 0 | εs | 0 | 0 | 0 |
| Vspg1* | Vppσg3* + Vppπg4* | 1/2(Vpp&sigma - Vppπ)g1* | 0 | 0 | εp | 0 | 0 |
| Vspg2* | 1/2(Vppσ - Vppπ)g1* | Vppσg4* + Vppπg3* | 0 | 0 | 0 | εp | 0 |
| 0 | 0 | 0 | Vppπg0* | 0 | 0 | 0 | εp |
The result of finding the eigenstates of this matrix is that the π- and π*-bands touch at the six K points in the first Brillouin zone. This is the origin of the metallic electronic behavior of graphene.
The primary difference between the electronic structure of the graphene sheet and a nanotube with chirality Ch is due to the finite circumference of the nanotube. Instead of a continuous range for k in the azimuthal direction, there is a quantization of allowed k due to the boundary condition imposed by Ch. The band structure for a carbon nanotube is thus comprized of several one-dimensional cross-sections (in the direction of longitudinal k) of the graphene band structure. If these cross-sections pass through the K symmetry points, the nanotube is metallic. If we know the chirality of a nanotube, we can predict whether or not it will be metallic. The relationship that places the 1D band cross-sections through the K points is given by:
| n1 + 2n2 = 3m |
This relationship implies families of chiralities, such as the "armchair" nanotubes, where n1 = n2, which are always metallic, and the "zig-zag", where n2 = 0, one-third of which are metallic. For chiralities that don't meet the above requirement, the nanotube is predicted to be a semiconductor, with the band gap given by
| Eg = (3.13eV) |d1| D-1 |
where D is the diameter of the nanotube and |d1| is the magnitude of the nearest-neighbor vector, i.e. the nearest-neighbor distance. Figure 2 shows the first Brillouin zone of graphene and the cross section lines for a metallic carbon nanotube with chirality (6,6). The cross sections for this chirality lie parallel to the vector (b1 - b2)/2. The spacing between the cross section lines is given by (b1 + b2)/12, as derived from expressions in the report by K. Ghosh [2].
The structure, i.e. chirality, of single-walled carbon nanotubes has been observed through resonant Raman scattering. An excellent reference for this topic is found in the Jorio Phys Rev Letters paper [3].
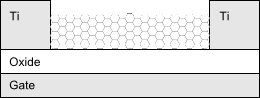 |
| Fig. 3: Setup used by H Dai to measure conductance of nanotubes. |
The electronic properties of single-walled nanotubes deposited by chemical vapor deposition have been found by transport measurements. It was observed that the majority of the nanotubes were semiconducting in nature with p-type behavior. In the same experiment, metallic (armchair) type nanotubes exhibited long mean free path ballistic electron transport and were minimally sensitive to the applied gate voltage. A third type of tube with band gaps around 10meV showed quasi-metallic behavior. This type of tube was not much affected by electrostatic doping but did exhibit a distinct conductance dip, due to the small band gap. The hypothesized explanation for this effect is a change in hybridization from the sp2 of graphene to sp3 due to the curvature of the nanotube. The metallic and quasi-metallic nanotubes approached a conductance of 4e2/h at low temperatures (1.5K). For more information on the electronic and other properties of nanotubes, the Acc. Chem. Res. review article by Hongjie Dai [4] is a good place to start.
It is evident through numerous observations that, although carbon nanotubes are largely inert, the electrical properties are sensitive to the effects of electron transfer and chemical doping. For example, the hole doping of semiconducting nanotubes does not occur in oxygen free environments. Intrinsic semiconductor behavior in vacuum was observed by Robert Chen et al[5].
It is clear that carbon nanotubes of different type are desirable for different applications. For example, the semiconductors might be used for future transistor applications while the metallic tubes could be used as nearly ideal 1D nanowires. However, since the nature of the deposition methods currently used produce both semiconducting and metallic tubes in an uncontrolled fashion, separation methods are needed until such time as the chirality of carbon nanotubes can be controlled in deposition. One method for separating metallic and semiconducting tubes relies on the difference in the induced dipoles of the two types of nanotubes. Krupe et al. [6] have demonstrated that separation occurs under AC dielectrophoresis. Their results showed that the nanotubes left on the microelectrodes were almost all metallic, while the proportion of metallic to semiconducting nanotubes in the remaining carrier solution was not significantly changed. An alternate approach developed by L. An et al. [7] removes the metallic nanotubes in a wafer-scale compatible process. In this setup, the metallic nanotubes are removed by repeated reaction with 4-bromobenzenediazonium.
© 2007 Jason D. Komadina. The author grants permission to copy, distribute and display this work in unaltered form, with attribution to the author, for noncommercial purposes only. All other rights, including commercial rights, are reserved to the author.
[1] E. Kaxiras, Atomic and Electronic Structure of Solids (Cambridge, 2003).
[2] K. Ghosh, "Electronic Band Structure of Carbon Nanotubes", coursework for EE228, Stanford University.
[3] A. Jorio et al., "Structural (n,m) Determination of Isolated Single-Wall Carbon Nanotubes by Resonant Raman Scattering", Phys. Rev. Lett. 86, 1118 (2001).
[4] H. Dai, "Carbon Nanotubes: Synthesis, Integration, and Properties", Acc. Chem. Res. 35, 1035 (2002).
[5] R. Chen et al., "Molecular photodesorption from single-walled carbon nanotubes", Appl. Phys. Lett. 79, 2258 (2001).
[6] R. Krupke et al., "Separation of Metallic from Semiconducting Single-Walled Carbon Nanotubes", Science 301, 344.
[6] L. An et al, "A Simple Chemical Route To Selectively Eliminate Metallic Carbon Nanotubes in Nanotube Network Devices", J. Am. Chem. Soc. 126, 10520 (2004).